Distribution of eQTL sharing across tissues
Sarah Urbut, Gao Wang, Peter Carbonetto and Matthew Stephens
Last updated: 2018-06-18
workflowr checks: (Click a bullet for more information)-
✔ R Markdown file: up-to-date
Great! Since the R Markdown file has been committed to the Git repository, you know the exact version of the code that produced these results.
-
✔ Environment: empty
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
-
✔ Seed:
set.seed(1)The command
set.seed(1)was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible. -
✔ Session information: recorded
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
-
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility. The version displayed above was the version of the Git repository at the time these results were generated.✔ Repository version: 14f28d4
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can usewflow_publishorwflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.Unstaged changes: Modified: analysis/hettablesim
Expand here to see past versions:
| File | Version | Author | Date | Message |
|---|---|---|---|---|
| Rmd | 6314ce0 | Gao Wang | 2018-06-16 | Relabel ‘test’ to ‘strong’ in data and code |
| html | 6eee6a9 | Peter Carbonetto | 2018-06-06 | Updated the webpages for a bunch of R Markdown files after minor revisions. |
| Rmd | 3dc0ba5 | Peter Carbonetto | 2018-06-06 | wflow_publish(c(“ExpressionAnalysis.Rmd”, “fastqtl2mash.Rmd”, |
| html | 64a343f | Peter Carbonetto | 2018-06-06 | Build site. |
| Rmd | 1534f7b | Peter Carbonetto | 2018-06-06 | wflow_publish(“SharingHist.Rmd”) |
| html | 5aaf7c2 | Peter Carbonetto | 2018-06-06 | Rebuilt page for revised SharingHist analysis. |
| Rmd | 1fcccc4 | Peter Carbonetto | 2018-06-06 | wflow_publish(“SharingHist.Rmd”) |
| html | e37a93d | Peter Carbonetto | 2018-06-06 | I have a first complete revision of the SharingHist analysis without |
| Rmd | dd35bf1 | Peter Carbonetto | 2018-06-06 | wflow_publish(“SharingHist.Rmd”) |
| Rmd | 35ca901 | Peter Carbonetto | 2018-06-06 | Revised data/results loading steps in HeterogeneityTables.Rmd. |
| Rmd | 0fa57df | Peter Carbonetto | 2018-06-06 | wflow_publish(“Tspecific.Rmd”) |
Here we develop another summary of sharing of eQTLs across tissues by showing the estimated distribution of sharing; specifically, the distribution of the number of tissues shared by sign and by magnitude. This complements this summary and this one.
Compare the plots below to the plots in Figure 5 of the paper.
Set up environment
First, we load some functions defined for the mash analyses.
source("../code/normfuncs.R")This is the threshold used to determine whether a gene has an eQTL in a given tissue.
thresh <- 0.05Load data and mash results
Load some GTEx summary statistics, as well as some of the results generated from the mash analysis of the GTEx data.
out <- readRDS("../data/MatrixEQTLSumStats.Portable.Z.rds")
maxbeta <- out$strong.b
maxz <- out$strong.z
out <- readRDS(paste("../output/MatrixEQTLSumStats.Portable.Z.coved.K3.P3",
"lite.single.expanded.V1.posterior.rds",sep = "."))
lfsr <- out$lfsr
pm.mash <- out$posterior.means
standard.error <- maxbeta/maxz
pm.mash.beta <- pm.mash * standard.errorHistograms of sharing by magnitude
Sharing by magnitude means that two eQTLs have similar effect size (within a factor of 2).
The histograms below summarize sharing by magnitude across all tissues (left), tissues other than brain (middle), and brain tissues only (right).
sigmat <- (lfsr <= thresh)
nsig <- rowSums(sigmat)
par(mar = c(4,4,2,1))
par(oma = c(8,4,0,0) + 0.1)
par(mfrow = c(1,3))
hist((het.func(het.norm(effectsize=pm.mash.beta[nsig>0,]),threshold=0.5)),
main="",xlab="",breaks=0.5:44.5,col="grey",freq=FALSE,ylim=c(0,0.9),
xaxt="n")
axis(1,at = seq(1, 44, by=1),labels = c(1:44))
mtext("All Tissues")
sigmat <- (lfsr[,-c(7:16)] <= thresh)
nsig <- rowSums(sigmat)
hist((het.func(het.norm(effectsize=pm.mash.beta[nsig>0,-c(7:16)]),
threshold=0.5)),main="",xlab="",breaks=0.5:34.5,col="grey",
freq=FALSE,ylim=c(0,0.9),xaxt="n")
axis(1,at = seq(1, 34, by=1),labels = c(1:34))
mtext("Non-Brain Tissues")
sigmat <- (lfsr[,c(7:16)]<=thresh)
nsig <- rowSums(sigmat)
brain.norm <- het.norm(effectsize=pm.mash.beta[nsig>0,c(7:16)])
hist(het.func(brain.norm,threshold=0.5),main="",xlab="",breaks=0.5:10.5,
col="grey",freq=FALSE,xaxt="n",ylim=c(0,0.9))
axis(1, at=seq(1, 10, by=1), labels=c(1:10))
mtext("Brain Tissues")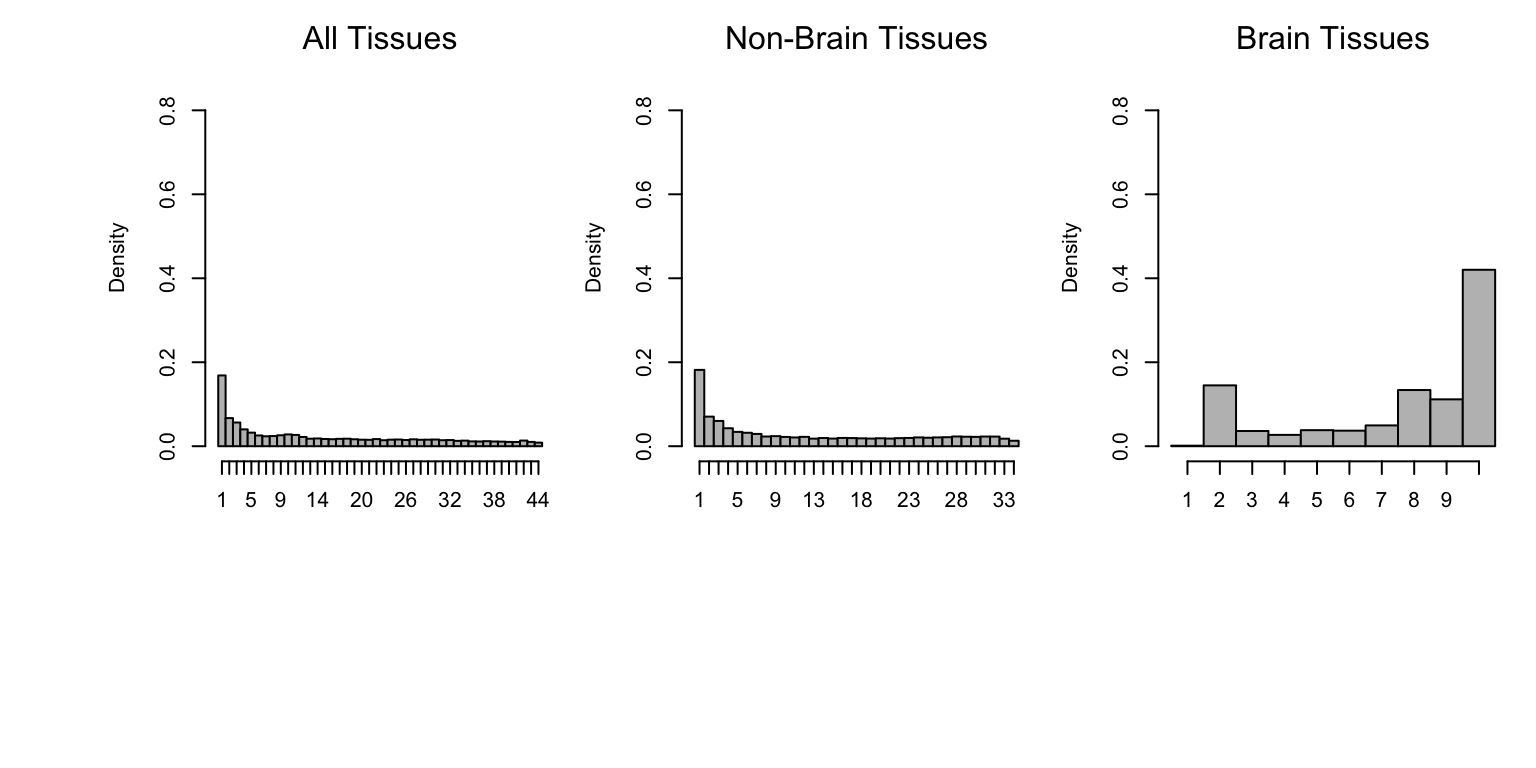
Expand here to see past versions of hindex-plot-magnitude-1.png:
| Version | Author | Date |
|---|---|---|
| e37a93d | Peter Carbonetto | 2018-06-06 |
Observe that the distribution of the number of tissues in which an eQTL is shared by magnitude has a mode at 1. This is a subset of eQTLs that have much stronger effect in one tissue than in any other (“tissue-specific”).
Histogram of sharing by magnitude
Sharing by sign means that the eQTLs have the same sign of effect.
The histograms below summarize sharing by sign across all tissues (left), tissues other than brain (middle), and brain tissues only (right).
sign.func <- function (normeffectsize)
apply(normeffectsize,1,function(x)(sum(x>0)))
sigmat <- (lfsr<=thresh)
nsig <- rowSums(sigmat)
par(mar = c(4,4.5,2,1))
par(oma = c(8,4,0,0) + 0.1)
par(mfrow = c(1,3))
hist(sign.func(het.norm(effectsize=pm.mash.beta[nsig>0,])),main="",xlab="",
breaks=0.5:44.5,col="grey",freq=FALSE,xaxt="n",ylim=c(0,0.9))
axis(1, at=seq(1, 44, by=1), labels=c(1:44))
mtext("All Tissues")
sigmat <- (lfsr[,-c(7:16)] <= thresh)
nsig <- rowSums(sigmat)
hist(sign.func(het.norm(effectsize = pm.mash.beta[nsig>0,-c(7:16)])),
main="",xlab="",breaks=0.5:34.5,col="grey",freq=FALSE,ylim=c(0,0.9),
xaxt="n")
axis(1, at=seq(1, 34, by=1), labels=c(1:34))
mtext("Non-Brain Tissues")
sigmat <- (lfsr[,c(7:16)]<=thresh)
nsig <- rowSums(sigmat)
brain.norm <- het.norm(effectsize=pm.mash.beta[nsig>0,c(7:16)])
hist(sign.func(brain.norm),main="",xlab="",breaks=0.5:10.5,col="grey",
freq=FALSE,xaxt="n",ylim=c(0,0.9))
axis(1, at=seq(1, 10, by=1), labels=c(1:10))
mtext("Brain Tissues")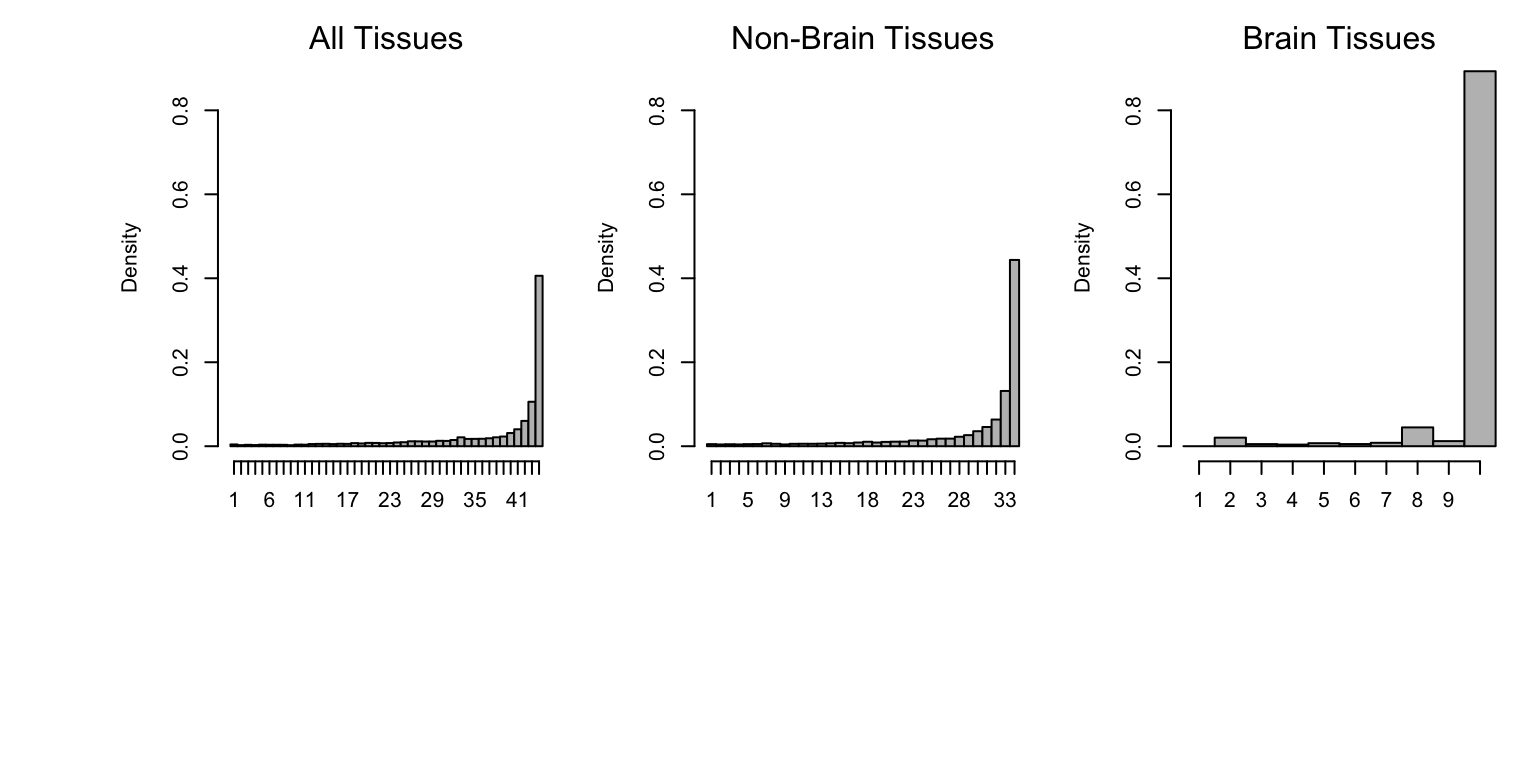
Expand here to see past versions of hindex-plot-sign-1.png:
| Version | Author | Date |
|---|---|---|
| e37a93d | Peter Carbonetto | 2018-06-06 |
Similar to other summaries of the mash analysis, we see that eQTLs exhibit a high level of sharing across tissues.
Session information
sessionInfo()
# R version 3.4.3 (2017-11-30)
# Platform: x86_64-apple-darwin15.6.0 (64-bit)
# Running under: macOS High Sierra 10.13.5
#
# Matrix products: default
# BLAS: /Library/Frameworks/R.framework/Versions/3.4/Resources/lib/libRblas.0.dylib
# LAPACK: /Library/Frameworks/R.framework/Versions/3.4/Resources/lib/libRlapack.dylib
#
# locale:
# [1] en_US.UTF-8/en_US.UTF-8/en_US.UTF-8/C/en_US.UTF-8/en_US.UTF-8
#
# attached base packages:
# [1] stats graphics grDevices utils datasets methods base
#
# loaded via a namespace (and not attached):
# [1] workflowr_1.0.1.9000 Rcpp_0.12.17 digest_0.6.15
# [4] rprojroot_1.3-2 R.methodsS3_1.7.1 backports_1.1.2
# [7] git2r_0.21.0 magrittr_1.5 evaluate_0.10.1
# [10] stringi_1.1.7 whisker_0.3-2 R.oo_1.21.0
# [13] R.utils_2.6.0 rmarkdown_1.9 tools_3.4.3
# [16] stringr_1.3.0 yaml_2.1.18 compiler_3.4.3
# [19] htmltools_0.3.6 knitr_1.20This reproducible R Markdown analysis was created with workflowr 1.0.1.9000