RTG Command Reference¶
This chapter describes RTG commands with a generic description of parameter options and usage. This section also includes expected operation and output results.
Command line interface (CLI)¶
RTG is installed as a single executable in any system subdirectory where permissions authorize a particular community of users to run the application. RTG commands are executed through the RTG command-line interface (CLI). Each command has its own set of parameters and options described in this section.
Results are organized in results directories defined by command
parameters and settings. The command line shell environment should
include a set of familiar text post-processing tools, such as grep,
awk, or perl. Otherwise, no additional applications such as
databases or directory services are required.
RTG command syntax¶
Usage:
rtg COMMAND [OPTIONS] <REQUIRED>
To run an RTG command at the command prompt (either DOS window or Unix terminal), type the product name followed by the command and all required and optional parameters. For example:
$ rtg format -o human_REF_SDF human_REF.fasta
Typically results are written to output files specified with the -o
option. There is no default filename or filename extension added to
commands requiring specification of an output directory or format.
Many times, unfiltered output files are very large; the built-in
compression option generates block compressed output files with the
.gz extension automatically unless the parameter -Z or --no-gzip
is issued with the command.
Many command parameters require user-supplied information of various types, as shown in the following:
Type |
Description |
|---|---|
DIR, FILE |
File or directory name(s) |
SDF |
Sequence data that has been formatted to SDF |
INT |
Integer value |
FLOAT |
Floating point decimal value |
STRING |
A sequence of characters for comments, filenames, or labels |
REGION |
A genomic region specification (see below) |
Genomic region parameters take one of the following forms:
sequence_name (e.g.:
chr21) corresponds to the entirety of the named sequence.sequence_name:start (e.g.:
chr21:100000) corresponds to a single position on the named sequence.sequence_name:start-end (e.g.:
chr21:100000-110000) corresponds to a range that extends from the specified start position to the specified end position (inclusive). The positions are 1-based.sequence_name:position+length (e.g.:
chr21:100000+10000) corresponds to a range that extends from the specified start position that includes the specified number of nucleotides.sequence_name:position~padding (e.g.:
chr21:100000~10000) corresponds to a range that spans the specified position by the specified amount of padding on either side.
To display all parameters and syntax associated with an RTG command,
enter the command and type --help. For example: all parameters
available for the RTG format command are displayed when rtg format
--help is executed, the output of which is shown below.
Usage: rtg format [OPTION]... -o SDF FILE+
[OPTION]... -o SDF -I FILE
[OPTION]... -o SDF -l FILE -r FILE
Converts the contents of sequence data files (FASTA/FASTQ/SAM/BAM) into the RTG
Sequence Data File (SDF) format.
File Input/Output
-f, --format=FORMAT format of input. Allowed values are [fasta,
fastq, sam-se, sam-pe, cg-fastq, cg-sam]
(Default is fasta)
-I, --input-list-file=FILE file containing a list of input read files (1
per line)
-l, --left=FILE left input file for FASTA/FASTQ paired end
data
-o, --output=SDF name of output SDF
-p, --protein input is protein. If this option is not
specified, then the input is assumed to
consist of nucleotides
-q, --quality-format=FORMAT format of quality data for fastq files (use
sanger for Illumina 1.8+). Allowed values are
[sanger, solexa, illumina]
-r, --right=FILE right input file for FASTA/FASTQ paired end
data
FILE+ input sequence files. May be specified 0 or
more times
Filtering
--duster treat lower case residues as unknowns
--exclude=STRING exclude input sequences based on their name.
If the input sequence contains the specified
string then that sequence is excluded from the
SDF. May be specified 0 or more times
--select-read-group=STRING when formatting from SAM/BAM input, only
include reads with this read group ID
--trim-threshold=INT trim read ends to maximise base quality above
the given threshold
Utility
--allow-duplicate-names disable checking for duplicate sequence names
-h, --help print help on command-line flag usage
--no-names do not include name data in the SDF output
--no-quality do not include quality data in the SDF output
--sam-rg=STRING|FILE file containing a single valid read group SAM
header line or a string in the form
"@RG\tID:READGROUP1\tSM:BACT_SAMPLE\tPL:ILLUMINA"
Required parameters are indicated in the usage display; optional parameters are listed immediately below the usage information in organized categories.
Use the double-dash when typing the full-word command option, as in
--output:
$ rtg format --output human_REF_SDF human_REF.fasta
Commonly used command options provide an abbreviated single-character
version of a full command parameter, indicated with only a single dash,
(Thus --output is the same as specifying the command option with the
abbreviated character -o):
$ rtg format -o human_REF human_REF.fasta
A set of utility commands are provided through the CLI: version,
license, and help. Start with these commands to familiarize yourself
with the software.
The rtg version command invokes the RTG software and triggers the
launch of RTG product commands, options, and utilities:
$ rtg version
It will display the version of the RTG software installed, RAM requirements, for example:
$rtg version
Product: RTG Core 3.5
Core Version: 6236f4e (2014-10-31)
RAM: 40.0GB of 47.0GB RAM can be used by rtg (84%)
License: No license file required
Contact: support@realtimegenomics.com
Patents / Patents pending:
US: 7,640,256, 13/129,329, 13/681,046, 13/681,215, 13/848,653,
13/925,704, 14/015,295, 13/971,654, 13/971,630, 14/564,810
UK: 1222923.3, 1222921.7, 1304502.6, 1311209.9, 1314888.7, 1314908.3
New Zealand: 626777, 626783, 615491, 614897, 614560
Australia: 2005255348, Singapore: 128254
Citation:
John G. Cleary, Ross Braithwaite, Kurt Gaastra, Brian S. Hilbush, Stuart
Inglis, Sean A. Irvine, Alan Jackson, Richard Littin, Sahar
Nohzadeh-Malakshah, Mehul Rathod, David Ware, Len Trigg, and Francisco
M. De La Vega. "Joint Variant and De Novo Mutation Identification on
Pedigrees from High-Throughput Sequencing Data." Journal of
Computational Biology. June 2014, 21(6): 405-419.
doi:10.1089/cmb.2014.0029.
(c) Real Time Genomics Inc, 2014
To see release status of commands you are licensed to use, type rtg license:
$rtg license
License: No license file required
Command name Licensed? Release Level
Data formatting:
format Licensed GA
sdf2fasta Licensed GA
sdf2fastq Licensed GA
Utility:
bgzip Licensed GA
index Licensed GA
extract Licensed GA
sdfstats Licensed GA
sdfsubset Licensed GA
sdfsubseq Licensed GA
mendelian Licensed GA
vcfstats Licensed GA
vcfmerge Licensed GA
vcffilter Licensed GA
vcfannotate Licensed GA
vcfsubset Licensed GA
vcfeval Licensed GA
pedfilter Licensed GA
pedstats Licensed GA
rocplot Licensed GA
version Licensed GA
license Licensed GA
help Licensed GA
To display all commands and usage parameters available to use with your
license, type rtg help:
$ rtg help
Usage: rtg COMMAND [OPTION]...
rtg RTG_MEM=16G COMMAND [OPTION]... (e.g. to set maximum memory use to 16 GB)
Type ``rtg help COMMAND`` for help on a specific command. The
following commands are available:
Data formatting:
format convert a FASTA file to SDF
cg2sdf convert Complete Genomics reads to SDF
sdf2fasta convert SDF to FASTA
sdf2fastq convert SDF to FASTQ
sdf2sam convert SDF to SAM/BAM
Read mapping:
map read mapping
mapf read mapping for filtering purposes
cgmap read mapping for Complete Genomics data
Protein search:
mapx translated protein search
Assembly:
assemble assemble reads into long sequences
addpacbio add Pacific Biosciences reads to an assembly
Variant detection:
calibrate create calibration data from SAM/BAM files
svprep prepare SAM/BAM files for sv analysis
sv find structural variants
discord detect structural variant breakends using discordant reads
coverage calculate depth of coverage from SAM/BAM files
snp call variants from SAM/BAM files
family call variants for a family following Mendelian inheritance
somatic call variants for a tumor/normal pair
population call variants for multiple potentially-related individuals
lineage call de novo variants in a cell lineage
avrbuild AVR model builder
avrpredict run AVR on a VCF file
cnv call CNVs from paired SAM/BAM files
Metagenomics:
species estimate species frequency in metagenomic samples
similarity calculate similarity matrix and nearest neighbor tree
Simulation:
genomesim generate simulated genome sequence
cgsim generate simulated reads from a sequence
readsim generate simulated reads from a sequence
readsimeval evaluate accuracy of mapping simulated reads
popsim generate a VCF containing simulated population variants
samplesim generate a VCF containing a genotype simulated from a population
childsim generate a VCF containing a genotype simulated as a child of two parents
denovosim generate a VCF containing a derived genotype containing de novo variants
samplereplay generate the genome corresponding to a sample genotype
cnvsim generate a mutated genome by adding CNVs to a template
Utility:
bgzip compress a file using block gzip
index create a tabix index
extract extract data from a tabix indexed file
sdfstats print statistics about an SDF
sdfsplit split an SDF into multiple parts
sdfsubset extract a subset of an SDF into a new SDF
sdfsubseq extract a subsequence from an SDF as text
sam2bam convert SAM file to BAM file and create index
sammerge merge sorted SAM/BAM files
samstats print statistics about a SAM/BAM file
samrename rename read id to read name in SAM/BAM files
mapxrename rename read id to read name in mapx output files
mendelian check a multi-sample VCF for Mendelian consistency
vcfstats print statistics from about variants contained within a VCF file
vcfmerge merge single-sample VCF files into a single multi-sample VCF
vcffilter filter records within a VCF file
vcfannotate annotate variants within a VCF file
vcfsubset create a VCF file containing a subset of the original columns
vcfeval evaluate called variants for agreement with a baseline variant set
pedfilter filter and convert a pedigree file
pedstats print information about a pedigree file
avrstats print statistics about an AVR model
rocplot plot ROC curves from vcfeval ROC data files
usageserver run a local server for collecting RTG command usage information
version print version and license information
license print license information for all commands
help print this screen or help for specified command
The help command will only list the commands at GA or beta release level.
To display help and syntax information for a specific command from the command line, type the command and then the –help option, as in:
$ rtg format --help
Note
The following commands are synonymous:
rtg help format and rtg format --help
See also
Refer to Installation and deployment for information about installing the RTG product executable.
Data Formatting Commands¶
format¶
Synopsis:
The format command converts the contents of sequence data files
(FASTA/FASTQ/SAM/BAM) into the RTG Sequence Data File (SDF) format. This
step ensures efficient processing of very large data sets, by organizing
the data into multiple binary files within a named directory. The same
SDF format is used for storing sequence data, whether it be genomic
reference, sequencing reads, protein sequences, etc.
Syntax:
Format one or more files specified from command line into a single SDF:
$ rtg format [OPTION] -o SDF FILE+
Format one or more files specified in a text file into a single SDF:
$ rtg format [OPTION] -o SDF -I FILE
Format mate pair reads into a single SDF:
$ rtg format [OPTION] -o SDF -l FILE -r FILE
Examples:
For FASTA (.fa) genome reference data:
$ rtg format -o maize_reference maize_chr*.fa
For FASTQ (.fq) sequence read data:
$ rtg format -f fastq -q sanger -o h1_reads -l h1_sample_left.fq -r h1_sample_right.fq
Parameters:
File Input/Output |
||
|---|---|---|
|
|
The format of the input file(s). Allowed values are [fasta, fastq, fastq-interleaved, sam-se, sam-pe] (Default is fasta). |
|
|
Specifies a file containing a list of sequence data files (one per line) to be converted into an SDF. |
|
|
The left input file for FASTA/FASTQ paired end data. |
|
|
The name of the output SDF. |
|
|
Set if the input consists of protein. If this option is not specified, then the input is assumed to consist of nucleotides. |
|
|
The format of the quality data for fastq format files. (Use sanger for Illumina1.8+). Allowed values are [sanger, solexa, illumina]. |
|
|
The right input file for FASTA/FASTQ paired end data. |
|
Specifies a sequence data file to be converted into an SDF. May be specified 0 or more times. |
|
Filtering |
||
|---|---|---|
|
Treat lower case residues as unknowns. |
|
|
Exclude individual input sequences based on their name. If the input sequence name contains the specified string then that sequence is excluded from the SDF. May be specified 0 or more times. |
|
|
Set to only include only reads with this read group ID when formatting from SAM/BAM files. |
|
|
Set to trim the read ends to maximise the base quality above the given threshold. |
|
Utility |
||
|---|---|---|
|
Set to disable duplicate name detection. |
|
|
|
Prints help on command-line flag usage. |
|
Do not include sequence names in the resulting SDF. |
|
|
Do not include sequence quality data in the resulting SDF. |
|
|
Specifies a file containing a single valid read group SAM header line or a string in the form |
|
Usage:
Formatting takes one or more input data files and creates a single SDF.
Specify the type of file to be converted, or allow default to FASTA
format. To aggregate multiple input data files, such as when formatting
a reference genome consisting of multiple chromosomes, list all files on
the command line or use the --input-list-file flag to specify a file
containing the list of files to process.
For input FASTA and FASTQ files which are compressed, they must have a
filename extension of .gz (for gzip compressed data) or .bz2 (for
bzip2 compressed data).
When formatting human reference genome data, it is recommended that the
resulting SDF be augmented with chromosome reference metadata, in order
to enable automatic sex-aware features during mapping and variant
calling. The format command will automatically recognize several
common human reference genomes and install a reference configuration
file. If your reference genome is not recognized, a configuration can be
manually adapted from one of the examples provided in the RTG
distribution and installed in the SDF directory. The reference
configuration is described in RTG reference file format.
When using FASTQ input files you must specify the quality format being
used as one of sanger, solexa or illumina. As of Illumina pipeline
version 1.8 and higher, quality values are encoded in Sanger format and
so should be formatted using --quality-format=sanger. Output from
earlier Illumina pipeline versions should be formatted using
--quality-format=illumina for Illumina pipeline versions starting with
1.3 and before 1.8, or --quality-format=solexa for Illumina pipeline
versions less than 1.3.
For FASTQ files that represent paired-end read data, indicate each side
respectively using the --left=FILE and --right=FILE flags.
Sometimes paired-end reads are represented in a single FASTQ file by
interleaving each side of the read. This type of input can be
formatted by specifying fastq-interleaved as the format type.
The mapx command maps translated DNA sequence data against a protein
reference. You must use the -p, --protein flag to format the protein
reference used by mapx.
Use the sam-se format for single end SAM/BAM input files and the
sam-pe format for paired end SAM/BAM input files. Note that if the
input SAM/BAM files are sorted in coordinate order (for example if they
have already been aligned to a reference), it is recommended that they
be shuffled before formatting, so that subsequent mapping is not biased
by processing reads in chromosome order. For example, a BAM file can be
shuffled using samtools collate as follows:
$ samtools collate -uOn 256 reads.bam tmp-prefix >reads_shuffled.bam
And this can be carried out on the fly during formatting using bash process redirection in order to reduce intermediate I/O, for example:
$ rtg format --format sam-pe <(samtools collate -uOn 256 reads.bam temp-prefix) ...
The SDF for a read set can contain a SAM read group which will be
automatically picked up from the input SAM/BAM files if they contain
only one read group. If the input SAM/BAM files contain multiple read
groups you must select a single read group from the SAM/BAM file to
format using the --select-read-group flag or specify a custom read
group with the --sam-rg flag. The --sam-rg flag can also be used to
add read group information to reads given in other input formats. The
SAM read group stored in an SDF will be automatically used during
mapping the reads it contains to provide tracking information in the
output BAM files.
The --trim-threshold flag can be used to trim poor quality read ends
from the input reads by inspecting base qualities from FASTQ input. If
and only if the quality of the final base of the read is less than the
threshold given, a new read length is found which maximizes the overall
quality of the retained bases using the following formula.
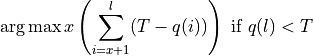
Where l is the original read length, x is the new read length, T is the given threshold quality and q(n) is the quality of the base at the position n of the read.
Note
Sequencing system read files and reference genome files often have the same extension and it may not always be obvious which file is a read set and which is a genome. Before formatting a sequencing system file, open it to see which type of file it is. For example:
$ less pf3.fa
In general, a read file typically begins with an @ or + character; a
genome reference file typically begins with the characters chr.
Normally when the input data contains multiple sequences with the same name the
format command will fail with an error. The --allow-duplicate-names flag
will disable this check conserving memory, but if the input data has multiple
sequences with the same name you will not be warned. Having duplicate sequence
names can cause problems with other commands, especially for reference data
since the output many commands identifies sequences by their names.
sdf2fasta¶
Synopsis:
Convert SDF data into a FASTA file.
Syntax:
$ rtg sdf2fasta [OPTION]... -i SDF -o FILE
Example:
$ rtg sdf2fasta -i humanSDF -o humanFASTA_return
Parameters:
File Input/Output |
||
|---|---|---|
|
|
SDF containing sequences. |
|
|
Output filename (extension added if not present). Use ‘-’ to write to standard output. |
Filtering |
||
|---|---|---|
|
Only output sequences with sequence id less than the given number. (Sequence ids start at 0). |
|
|
Only output sequences with sequence id greater than or equal to the given number. (Sequence ids start at 0). |
|
|
|
Name of a file containing a list of sequences to extract, one per line. |
|
Interpret any specified sequence as names instead of numeric sequence ids. |
|
|
Interpret any specified sequence as taxon ids instead of numeric sequence ids. This option only applies to a metagenomic reference species SDF. |
|
|
Specify one or more explicit sequences to extract, as sequence id, or sequence name if –names flag is set. |
|
Utility |
||
|---|---|---|
|
|
Prints help on command-line flag usage. |
|
Interleave paired data into a single output file. Default is to split to separate output files. |
|
|
|
Set the maximum number of nucleotides or amino acids to print on a line of FASTA output. Should be nonnegative, with a value of 0 indicating that the line length is not capped. (Default is 0). |
|
|
Set this flag to create the FASTA output file without compression. By default the output file is compressed with blocked gzip. |
Usage:
Use the sdf2fasta command to convert SDF data into FASTA format. By
default, sdf2fasta creates a separate line of FASTA output for each
sequence. These lines will be as long as the sequences themselves. To
make them more readable, use the -l, --line-length flag and define a
reasonable record length like 75.
By default all sequences will be extracted, but flags may be specified
to extract reads within a range, or explicitly specified reads (either
by numeric sequence id or by sequence name if --names is set).
Additionally, when the input SDF is a metagenomic species reference SDF,
the --taxons option, any supplied id is interpreted as a taxon id and
all sequences assigned directly to that taxon id will be output. This
provides a convenient way to extract all sequence data corresponding to
a single (or multiple) species from a metagenomic species reference SDF.
Sequence ids are numbered starting at 0, the --start-id flag is an inclusive
lower bound on id and the --end-id flag is an exclusive upper bound. For
example if you have an SDF with five sequences (ids: 0, 1, 2, 3, 4) the
following command:
$ rtg sdf2fasta --start-id=3 -i mySDF -o output
will extract sequences with id 3 and 4. The command:
$ rtg sdf2fasta --end-id=3 -i mySDF -o output
will extract sequences with id 0, 1, and 2. And the command:
$ rtg sdf2fasta --start-id=2 --end-id=4 -i mySDF -o output
will extract sequences with id 2 and 3.
sdf2fastq¶
Synopsis:
Convert SDF data into a FASTQ file.
Syntax:
$ rtg sdf2fastq [OPTION]... -i SDF -o FILE
Example:
$ rtg sdf2fastq -i humanSDF -o humanFASTQ_return
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Specifies the SDF data to be converted. |
|
|
Specifies the file name used to write the resulting FASTQ output. |
Filtering |
||
|---|---|---|
|
Only output sequences with sequence id less than the given number. (Sequence ids start at 0). |
|
|
Only output sequences with sequence id greater than or equal to the given number. (Sequence ids start at 0). |
|
|
|
Name of a file containing a list of sequences to extract, one per line. |
|
Interpret any specified sequence as names instead of numeric sequence ids. |
|
|
Specify one or more explicit sequences to extract, as sequence id, or sequence name if –names flag is set. |
|
Utility |
||
|---|---|---|
|
|
Prints help on command-line flag usage. |
|
|
Set the default quality to use if the SDF does not contain sequence quality data (0-63). |
|
Interleave paired data into a single output file. Default is to split to separate output files. |
|
|
|
Set the maximum number of nucleotides or amino acids to print on a line of FASTQ output. Should be nonnegative, with a value of 0 indicating that the line length is not capped. (Default is 0). |
|
|
Set this flag to create the FASTQ output file without compression. By default the output file is compressed with blocked gzip. |
Usage:
Use the sdf2fastq command to convert SDF data into FASTQ format. If no
quality data is available in the SDF, use the -q, --default-quality
flag to set a quality score for the FASTQ output. The quality encoding
used during output is sanger quality encoding. By default, sdf2fastq
creates a separate line of FASTQ output for each sequence. As with
sdf2fasta, there is an option to use the -l, --line-length flag to
restrict the line lengths to improve readability of long sequences.
By default all sequences will be extracted, but flags may be specified
to extract reads within a range, or explicitly specified reads (either
by numeric sequence id or by sequence name if --names is set).
It may be preferable to extract data to unaligned SAM/BAM format using
sdf2sam, as this preserves read-group information stored in the SDF
and may also be more convenient when dealing with paired-end data.
The --start-id and --end-id flags behave as in sdf2fasta.
sdf2sam¶
Synopsis:
Convert SDF read data into unaligned SAM or BAM format file.
Syntax:
$ rtg sdf2sam [OPTION]... -i SDF -o FILE
Example:
$ rtg sdf2sam -i samplereadsSDF -o samplereads.bam
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Specifies the SDF data to be converted. |
|
|
Specifies the file name used to write the resulting SAM/BAM to. The output format is automatically determined based on the filename specified. If ‘-’ is given, the data is written as uncompressed SAM to standard output. |
Filtering |
||
|---|---|---|
|
Only output sequences with sequence id less than the given number. (Sequence ids start at 0). |
|
|
Only output sequences with sequence id greater than or equal to the given number. (Sequence ids start at 0). |
|
|
|
Name of a file containing a list of sequences to extract, one per line. |
|
Interpret any specified sequence as names instead of numeric sequence ids. |
|
|
Specify one or more explicit sequences to extract, as sequence id, or sequence name if –names flag is set. |
|
Utility |
||
|---|---|---|
|
|
Prints help on command-line flag usage. |
|
|
Set this flag when creating SAM format output to disable compression. By default SAM is compressed with blocked gzip, and BAM is always compressed. |
Usage:
Use the sdf2sam command to convert SDF data into unaligned SAM/BAM
format. By default all sequences will be extracted, but flags may be
specified to extract reads within a range, or explicitly specified reads
(either by numeric sequence id or by sequence name if --names is set).
This command is a useful way to export paired-end data to a single
output file while retaining any read group information that may be
stored in the SDF.
The output format is either SAM/BAM depending on the specified output file name.
e.g. output.sam or output.sam.gz will output as SAM, whereas
output.bam will output as BAM. If neither SAM or BAM format is indicated by
the file name then BAM will be used and the output file name adjusted
accordingly. e.g output will become output.bam. However if standard
output is selected (-) then the output will always be in uncompressed SAM
format.
The --start-id and --end-if behave as in sdf2fasta.
fastqtrim¶
Synopsis:
Trim reads in FASTQ files.
Syntax:
$ rtg fastqtrim [OPTION]... -i FILE -o FILE
Example:
Apply hard base removal from the start of the read and quality-based trimming of terminal bases:
$ rtg fastqtrim -s 12 -E 18 -i S12_R1.fastq.gz -o S12_trimmed_R1.fastq.gz
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Input FASTQ file, Use ‘-’ to read from standard input. |
|
|
Output filename. Use ‘-’ to write to standard output. |
|
|
Quality data encoding method used in FASTQ input files (Illumina 1.8+ uses sanger). Allowed values are [sanger, solexa, illumina] (Default is sanger) |
Filtering |
||
|---|---|---|
|
Discard reads that have zero length after trimming. Should not be used with paired-end data. |
|
|
|
Trim read ends to maximise base quality above the given threshold (Default is 0) |
|
If a read ends up shorter than this threshold it will be trimmed to zero length (Default is 0) |
|
|
|
Trim read starts to maximise base quality above the given threshold (Default is 0) |
|
|
Always trim the specified number of bases from read end (Default is 0) |
|
|
Always trim the specified number of bases from read start (Default is 0) |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
|
If set, output in reverse complement. |
|
Seed used during subsampling. |
|
|
If set, subsample the input to retain this fraction of reads. |
|
|
|
Number of threads (Default is the number of available cores) |
Usage:
Use fastqtrim to apply custom trimming and preprocessing to raw
FASTQ files prior to mapping and alignment. The format command
contains some limited trimming options, which are applied to all input
files, however in some cases different or specific trimming operations
need to be applied to the various input files. For example, for
paired-end data, different trimming may need to be applied for the left
read files compared to the right read files. In these cases,
fastqtrim should be used to process the FASTQ files first.
The --end-quality-threshold flag can be used to trim poor quality bases
from the ends of the input reads by inspecting base qualities from FASTQ
input. If and only if the quality of the final base of the read is less
than the threshold given, a new read length is found which maximizes the
overall quality of the retained bases using the following formula:
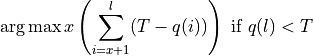
where l is the original read length, x is the new read length, T
is the given threshold quality and q(n) is the quality of the base at
the position n of the read. Similarly, --start-quality-threshold
can be used to apply this quality-based thresholding to the start of
reads.
Some of the trimming options may result in reads that have no bases
remaining. By default, these are output as zero-length FASTQ reads,
which RTG commands are able to handle normally. It is also possible to
remove zero-length reads altogether from the output with the
--discard-empty-reads option, however this should not be used when
processing FASTQ files corresponding to paired-end data, otherwise the
pairs in the two files will no longer be matched.
Similarly, when using the --subsample option to down-sample a FASTQ
file for paired-end data, you should specify an explicit randomization
seed via --seed and use the same seed value for the left and right
files.
Formatting with filtering on the fly¶
Running custom filtering with fastqtrim need not mean that
additional disk space is required or that formatting be slowed down due
to additional disk I/O. It is possible when using standard unix shells
to perform the filtering on the fly. The following example demonstrates
how to apply different trimming options to left and right files while
formatting to SDF:
$ rtg format -f fastq -o S12_trimmed.sdf \
-l <(rtg fastqtrim -s 12 -E 18 -i S12_R1.fastq.gz -o -)
-r <(rtg fastqtrim -E 18 -i S12_R2.fastq.gz -o -)
See also
petrim¶
Synopsis:
Trim paired-end read FASTQ files based on read arm alignment overlap.
Syntax:
$ rtg petrim [OPTION]... -l FILE -o FILE -r FILE
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Left input FASTQ file (AKA R1) |
|
|
Output filename prefix. Use ‘-’ to write to standard output. |
|
|
Quality data encoding method used in FASTQ input files (Illumina 1.8+ uses sanger). Allowed values are [sanger, solexa, illumina] (Default is sanger) |
|
|
Right input FASTQ file (AKA R2) |
Sensitivity Tuning |
||
|---|---|---|
|
Aligner indel band width scaling factor, fraction of read length allowed as an indel (Default is 0.5) |
|
|
Penalty for a gap extension during alignment (Default is 1) |
|
|
Penalty for a gap open during alignment (Default is 19) |
|
|
|
Minimum percent identity in overlap to trigger overlap trimming (Default is 90) |
|
|
Minimum number of bases in overlap to trigger overlap trimming (Default is 25) |
|
Penalty for a mismatch during alignment (Default is 9) |
|
|
Soft clip alignments if indels occur INT bp from either end (Default is 5) |
|
|
Penalty for unknown nucleotides during alignment (Default is 5) |
|
Filtering |
||
|---|---|---|
|
If set, discard pairs where both reads have zero length (after any trimming) |
|
|
If set, discard pairs where either read has zero length (after any trimming) |
|
|
Assume R1 starts with probes this long, and trim R2 bases that overlap into this (Default is 0) |
|
|
|
If set, merge overlapping reads at midpoint of overlap region. Result is in R1 (R2 will be empty) |
|
|
If set, trim overlapping reads to midpoint of overlap region. |
|
If a read ends up shorter than this threshold it will be trimmed to zero length (Default is 0) |
|
|
Method used to alter bases/qualities at mismatches within overlap region. Allowed values are [none, zero-phred, pick-best] (Default is none) |
|
|
Assume R2 starts with probes this long, and trim R1 bases that overlap into this (Default is 0) |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
Interleave paired data into a single output file. Default is to split to separate output files. |
|
|
|
Do not gzip the output. |
|
Seed used during subsampling. |
|
|
If set, subsample the input to retain this fraction of reads. |
|
|
|
Number of threads (Default is the number of available cores) |
Usage:
Paired-end read sequencing with read lengths that are long relative to the typical library fragment size can often result in the same bases being sequenced by both arms. This repeated sequencing of bases within the same fragment can skew variant calling, and so it can be advantageous to remove such read overlap.
In some cases, complete read-through can occur, resulting in additional adaptor or non-genomic bases being present at the ends of reads.
In addition, some library preparation methods rely on the ligation of
synthetic probe sequence to attract target DNA, which is subsequently
sequenced. Since these probe bases do not represent genomic material,
they must be removed at some point during the analytic pipeline prior to
variant calling, otherwise they could act as a reference bias when
calling variants. Removal from the primary arm where the probe is
attached is typically easy enough (e.g. via fastqtrim), however in
cases of high read overlap, probe sequence can also be present in the
other read arm.
petrim aligns each read arm against it’s mate with high stringency
in order to identify cases of read overlap. The sensitivity of read
overlap detection is primarily controlled through the use of
--min-identity and --min-overlap-length, although it is also
possible to adjust the penalties used during alignment.
The following types of trimming or merging may be applied.
Removal of non-genomic bases due to complete read-through. This removal is always applied.
Removal of overlap bases impinging into regions occupied by probe bases. For example, if the left arms contain 11-mer probes, using
--left-probe-length=11will result in the removal of any right arm bases that overlap into the first 11 bases of the left arm. Similar trimming is available for situations where probes are ligated to the right arm by using--right-probe-length.Adjustment of mismatching read bases inside areas of overlap. Such mismatches indicate that one or other of the bases has been incorrectly sequenced. Alteration of these bases is selected by supplying the
--mismatch-adjustmentflag with a value ofzero-phredto alter the phred quality score of both bases to zero, orpick-bestto choose whichever base had the higher reported quality score.Removal of overlap regions by trimming both arms back to a point where no overlap is present. An equal number of bases are removed from each arm. This trimming is enabled by specifying
--midpoint-trimand takes place after any read-through or probe related trimming.Merging non-redundant sequence from both reads to create a single read, enabled via
--midpoint-merge. This is like--midpoint-trimwith a subsequent moving of the R2 read onto the end of the the R1 read (thus the R2 read becomes empty).
After trimming or merging it is possible that one or both of the arms of
the pair have no bases remaining, and a strategy is needed to handle
these pairs. The default is to retain such pairs in the output, even if
one or both are zero-length. When both arms are zero-length, the pair
can be dropped from output with the use of --discard-empty-pairs. If
downstream processing cannot handle zero-length reads,
--discard-empty-reads will drop a read pair if either of the arms is
zero-length.
petrim also provides the ability to down-sample a read set by using
the --subsample option. This will produce a different sampling each time,
unless an explicit randomization seed is specified via --seed.
Formatting with paired-end trimming on the fly¶
Running custom filtering with petrim can be done in standard Unix
shells without incurring the use of additional disk space or unduly
slowing down the formatting of reads. The following example demonstrates
how to apply paired-end trimming while formatting to SDF:
$ rtg format -f fastq-interleaved -o S12_trimmed.sdf \
<(rtg petrim -l S12_R1.fastq.gz -r S12_R2.fastq.gz -m -o - --interleaved)
This can even be combined with fastqtrim to provide extremely
flexible trimming:
$ rtg format -f fastq-interleaved -o S12_trimmed.sdf \
<(rtg petrim -m -o - --interleave \
-l <(rtg fastqtrim -s 12 -E 18 -i S12_R1.fastq.gz -o -) \
-r <(rtg fastqtrim -E 18 -i S12_R2.fastq.gz -o -) \
)
Note
petrim currently assumes Illumina paired-end sequencing,
and aligns the reads in FR orientation. Sequencing methods which
produce arms in a different orientation can be processed by first
converting the input files using fastqtrim --reverse-complement,
running petrim, followed by another fastqtrim
--reverse-complement to restore the reads to their original
orientation.
Simulation Commands¶
RTG includes some simulation commands that may be useful for experimenting with effects of various RTG command parameters or when getting familiar with RTG work flows. A simple simulation series might involve the following commands:
$ rtg genomesim --output sim-ref-sdf --min-length 500000 --max-length 5000000 \
--num-contigs 5
$ rtg popsim --reference sim-ref-sdf --output population.vcf.gz
$ rtg samplesim --input population.vcf.gz --output sample1.vcf.gz \
--output-sdf sample1-sdf --reference sim-ref-sdf --sample sample1
$ rtg readsim --input sample1-sdf --output reads-sdf --machine illumina_pe \
-L 75 -R 75 --coverage 10
$ rtg map --template sim-ref-sdf --input reads-sdf --output sim-mapping \
--sam-rg "@RG\tID:sim-rg\tSM:sample1\tPL:ILLUMINA"
$ rtg snp --template sim-ref-sdf --output sim-name-snp sim-mapping/alignments.bam
genomesim¶
Synopsis:
Use the genomesim command to simulate a reference genome, or to create
a baseline reference genome for a research project when an actual genome
reference sequence is unavailable.
Syntax:
Specify number of sequences, plus minimum and maximum lengths:
$ rtg genomesim [OPTION]... -o SDF --max-length INT --min-length INT -n INT
Specify explicit sequence lengths (one more sequences):
$ rtg genomesim [OPTION]... -o SDF -l INT
Example:
$ rtg genomesim -o genomeTest -l 500000
Parameters:
File Input/Output |
||
|---|---|---|
|
|
The name of the output SDF. |
Utility |
||
|---|---|---|
|
Specify a comment to include in the generated SDF. |
|
|
Set the relative frequencies of A,C,G,T in the generated sequence. (Default is 1,1,1,1). |
|
|
|
Prints help on command-line flag usage. |
|
|
Specify the length of generated sequence. May be specified 0 or more times, or as a comma separated list. |
|
Specify the maximum sequence length. |
|
|
Specify the minimum sequence length. |
|
|
|
Specify the number of sequences to generate. |
|
Specify a sequence name prefix to be used for the generated sequences. The default is to name the output sequences ‘simulatedSequenceN’, where N is increasing for each sequence. |
|
|
|
Specify seed for the random number generator. |
Usage:
The genomesim command allows one to create a simulated genome with one
or more contiguous sequences - exact lengths of each contig or number of
contigs with minimum and maximum lengths provided. The contents of an
SDF directory created by genomesim can be exported to a FASTA file
using the sdf2fasta command.
This command is primarily useful for providing a simple randomly generated base genome for use with subsequent simulation commands.
Each generated contig is named by appending an increasing numeric index
to the specified prefix. For example --prefix=chr --num-contigs=10
would yield contigs named chr1 through chr10.
cgsim¶
Synopsis:
Simulate Complete Genomics Inc sequencing reads. Supports the original 35 bp read structure (5-10-10-10), and the newer 29 bp read structure (10-9-10).
Syntax:
Generation by genomic coverage multiplier:
$ rtg cgsim [OPTION]... -V INT -t SDF -o SDF -c FLOAT
Generation by explicit number of reads:
$ rtg cgsim [OPTION]... -V INT -t SDF -o SDF -n INT
Example:
$ rtg cgsim -V 1 -t HUMAN_reference -o CG_3x_readst -c 3
Parameters:
File Input/Output |
||
|---|---|---|
|
|
SDF containing input genome. |
|
|
Name for reads output SDF. |
Fragment Generation |
||
|---|---|---|
|
If set, the user-supplied distribution represents desired abundance. |
|
|
|
Allow reads to be drawn from template fragments containing unknown nucleotides. |
|
|
Coverage, must be positive. |
|
|
File containing probability distribution for sequence selection. |
|
If set, the user-supplied distribution represents desired DNA fraction. |
|
|
|
Maximum fragment size (Default is 500) |
|
|
Minimum fragment size (Default is 350) |
|
Rate that the machine will generate new unknowns in the read (Default is 0.0) |
|
|
|
Number of reads to be generated. |
|
File containing probability distribution for sequence selection expressed by taxonomy id. |
|
Complete Genomics |
||
|---|---|---|
|
|
Select Complete Genomics read structure version, 1 (35 bp) or 2 (29 bp) |
Utility |
||
|---|---|---|
|
Comment to include in the generated SDF. |
|
|
|
Print help on command-line flag usage. |
|
Do not create read names in the output SDF. |
|
|
Do not create read qualities in the output SDF. |
|
|
|
Set the range of base quality values permitted e.g.: 3-40 (Default is fixed qualities corresponding to overall machine base error rate) |
|
File containing a single valid read group SAM header line or a string in the form |
|
|
|
Seed for random number generator. |
Usage:
Use the cgsim command to set either --coverage or --num-reads in
simulated Complete Genomics reads. For more information about Complete
Genomics reads, refer to http://www.completegenomics.com
RTG simulation tools allows for deterministic experiment repetition. The
--seed parameter, for example, allows for regeneration of exact same
reads by setting the random number generator to be repeatable (without
supplying this flag a different set of reads will be generated each
time).
The --distribution parameter allows you to specify the probability
that a read will come from a particular named sequence for use with
metagenomic databases. Probabilities are numbers between zero and one
and must sum to one in the file.
readsim¶
Synopsis:
Use the readsim command to generate single or paired end reads of
fixed or variable length from a reference genome, introducing machine
errors.
Syntax:
Generation by genomic coverage multiplier:
$ rtg readsim [OPTION]... -t SDF --machine STRING -o SDF -c FLOAT
Generation by explicit number of reads:
$ rtg readsim [OPTION]... -t SDF --machine STRING -o SDF -n INT
Example:
$ rtg readsim -t genome_ref -o sim_reads -r 75 --machine illumina_se -c 30
Parameters:
File Input/Output |
||
|---|---|---|
|
|
SDF containing input genome. |
|
Select the sequencing technology to model. Allowed values are [illumina_se, illumina_pe, complete_genomics, complete_genomics_2, 454_pe, 454_se, iontorrent] |
|
|
|
Name for reads output SDF. |
Fragment Generation |
||
|---|---|---|
|
If set, the user-supplied distribution represents desired abundance. |
|
|
|
Allow reads to be drawn from template fragments containing unknown nucleotides. |
|
|
Coverage, must be positive. |
|
|
File containing probability distribution for sequence selection. |
|
If set, the user-supplied distribution represents desired DNA fraction. |
|
|
|
Maximum fragment size (Default is 250) |
|
|
Minimum fragment size (Default is 200) |
|
Rate that the machine will generate new unknowns in the read (Default is 0.0) |
|
|
|
Number of reads to be generated. |
|
File containing probability distribution for sequence selection expressed by taxonomy id. |
|
Illumina PE |
||
|---|---|---|
|
|
Target read length on the left side. |
|
|
Target read length on the right side. |
Illumina SE |
||
|---|---|---|
|
|
Target read length, must be positive. |
454 SE/PE |
||
|---|---|---|
|
Maximum 454 read length (in paired end case the sum of the left and the right read lengths) |
|
|
Minimum 454 read length (in paired end case the sum of the left and the right read lengths) |
|
IonTorrent SE |
||
|---|---|---|
|
Maximum IonTorrent read length. |
|
|
Minimum IonTorrent read length. |
|
Utility |
||
|---|---|---|
|
Comment to include in the generated SDF. |
|
|
|
Print help on command-line flag usage. |
|
Do not create read names in the output SDF. |
|
|
Do not create read qualities in the output SDF. |
|
|
|
Set the range of base quality values permitted e.g.: 3-40 (Default is fixed qualities corresponding to overall machine base error rate) |
|
File containing a single valid read group SAM header line or a string in the form |
|
|
|
Seed for random number generator. |
Usage:
Create simulated reads from a reference genome by either specifying coverage depth or a total number of reads.
A typical use case involves creating a mutated genome by introducing
SNPs or CNVs with popsim and samplesim generating reads from the
mutated genome with readsim, and mapping them back to the original
reference to verify the parameters used for mapping or variant
detection.
RTG simulation tools allows for deterministic experiment repetition. The
--seed parameter, for example, allows for regeneration of exact same
reads by setting the random number generator to be repeatable (without
supplying this flag a different set of reads will be generated each
time).
The --distribution parameter allows you to specify the sequence
composition of the resulting read set, primarily for use with
metagenomic databases. The distribution file is a text file containing
lines of the form:
<probability><space><sequence name>
Probabilities must be between zero and one and must sum to one in the
file. For reference databases containing taxonomy information, where
each species may be comprised of more than one sequence, it is instead
possible to use the --taxonomy-distribution option to specify the
probabilities at a per-species level. The format of each line in this
case is:
<probability><space><taxon id>
When using --distribution or --taxonomy-distribution, the
interpretation must be specified one of --abundance or
--dna-fraction. When using --abundance each specified
probability reflects the chance of selecting the specified sequence
(or taxon id) from the set of sequences, and thus for a given abundance
a large sequence will be represented by more reads in the resulting set
than a short sequence. In contrast, with --dna-fraction each
specified probability reflects the chance of a read being derived
from the designated sequence, and thus for a given fraction, a large
sequence will have a lower depth of coverage than a short sequence.
popsim¶
Synopsis:
Use the popsim command to generate a VCF containing simulated
population variants. Each variant allele generated has an associated
frequency INFO field describing how frequent in the population that
allele is.
Syntax:
$ rtg popsim [OPTION]... -o FILE -t SDF
Example:
$ rtg popsim -o pop.vcf -t HUMAN_reference
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Output VCF file name. |
|
|
SDF containing the reference genome. |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Seed for the random number generator. |
|
Usage:
The popsim command is used to create a VCF containing variants with
frequency in population information that can be subsequently used to
simulate individual samples using the samplesim command. The frequency
in population is contained in a VCF INFO field called AF. The types
of variants and the allele-frequency distribution has been drawn from
observed variants and allele frequency distribution in human studies.
See also
samplesim¶
Synopsis:
Use the samplesim command to generate a VCF containing a genotype
simulated from population variants according to allele frequency.
Syntax:
$ rtg samplesim [OPTION]... -i FILE -o FILE -t SDF -s STRING
Example:
From a population frequency VCF:
$ rtg samplesim -i pop.vcf -o 1samples.vcf -t HUMAN_reference -s person1 --sex male
From an existing simulated VCF:
$ rtg samplesim -i 1samples.vcf -o 2samples.vcf -t HUMAN_reference -s person2 \
--sex female
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Input VCF containing population variants. |
|
|
Output VCF file name. |
|
If set, output an SDF containing the sample genome. |
|
|
|
SDF containing the reference genome. |
|
|
Name for sample. |
Utility |
||
|---|---|---|
|
If set, treat variants without allele frequency annotation as uniformly likely. |
|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Ploidy to use. Allowed values are [auto, diploid, haploid] (Default is auto) |
|
|
Seed for the random number generator. |
|
|
Sex of individual. Allowed values are [male, female, either] (Default is either) |
|
Usage:
The samplesim command is used to simulate an individuals genotype
information from a population variant frequency VCF generated by the
popsim command or by previous samplesim or childsim commands. The
new output VCF will contain all the existing variants and samples with a
new column for the new sample. The genotype at each record of the VCF
will be chosen randomly according to the allele frequency specified in
the AF field.
If input VCF records do not contain an AF annotation, by default any
ALT allele in that record will not be selected and so the sample will be
genotyped as 0/0. Alternatively for simple simulations the
--allow-missing-af flag will treat each allele in such records as
being equally likely (i.e.: effectively equivalent to AF=0.5 for a
biallelic variant, AF=0.33,0.33 for a triallelic variant, etc).
The ploidy for each genotype is automatically determined according to
the ploidy of that chromosome for the specified sex of the individual,
as defined in the reference genome reference.txt file. For more
information see RTG reference file format. If the reference SDF
does not contain chromosome configuration information, a default ploidy
can be specified using the --ploidy flag.
The --output-sdf flag can be used to optionally generate an SDF of the
individuals genotype which can then be used by the readsim command to
simulate a read set for the individual.
See also
denovosim¶
Synopsis:
Use the denovosim command to generate a VCF containing a derived
genotype containing de novo variants.
Syntax:
$ rtg denovosim [OPTION]... -i FILE --original STRING -o FILE -t SDF -s STRING
Example:
$ rtg denovosim -i sample.vcf --original personA -o 2samples.vcf \
-t HUMAN_reference -s personB
Parameters:
File Input/Output |
||
|---|---|---|
|
|
The input VCF containing parent variants. |
|
The name of the existing sample to use as the original genotype. |
|
|
|
The output VCF file name. |
|
Set to output an SDF of the genome generated. |
|
|
|
The SDF containing the reference genome. |
|
|
The name for the new derived sample. |
Utility |
||
|---|---|---|
|
|
Prints help on command-line flag usage. |
|
|
Set this flag to create the VCF output file without compression. |
|
Set the expected number of de novo mutations per genome (Default is 70). |
|
|
The ploidy to use when the reference genome does not contain a reference text file. Allowed values are [auto, diploid, haploid] (Default is auto) |
|
|
Set the seed for the random number generator. |
|
|
Set this flag to display information regarding de novo mutation points. |
|
Usage:
The denovosim command is used to simulate a derived genotype
containing de novo variants from a VCF containing an existing
genotype.
The output VCF will contain all the existing variants and samples, along
with additional de novo variants. If the original and derived sample
names are different, the output will contain a new column for the
mutated sample. If the original and derived sample names are the same,
the sample in the output VCF is updated rather than creating an entirely
new sample column. When a sample receives a de novo mutation, the
sample DN field is set to “Y”.
If de novo variants were introduced without regard to neighboring
variants, a situation could arise where it is not possible to
unambiguously determine the haplotype of the simulated sample. To
prevent this, denovosim will not output a de novo variant that
overlaps existing variants. Since denovosim chooses candidate de
novo locations before reading the input VCF, this occasionally mandates
skipping a candidate de novo so the target number of mutations may not
always be reached.
The --output-sdf flag can be used to optionally generate an SDF of the
derived genome which can then be used by the readsim command to
simulate a read set for the new genome.
See also
childsim¶
Synopsis:
Use the childsim command to generate a VCF containing a genotype
simulated as a child of two parents.
Syntax:
$ rtg childsim [OPTION]... --father STRING -i FILE --mother STRING -o FILE -t SDF \
-s STRING
Example:
$ rtg childsim --father person1 --mother person2 -i 2samples.vcf -o 3samples.vcf \
-t HUMAN_reference -s person3
Parameters:
File Input/Output |
||
|---|---|---|
|
Name of the existing sample to use as the father. |
|
|
|
Input VCF containing parent variants. |
|
Name of the existing sample to use as the mother. |
|
|
|
Output VCF file name. |
|
If set, output an SDF containing the sample genome. |
|
|
|
SDF containing the reference genome. |
|
|
Name for new child sample. |
Utility |
||
|---|---|---|
|
Probability of extra crossovers per chromosome (Default is 0.01) |
|
|
If set, load genetic maps from this directory for recombination point selection. |
|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Ploidy to use. Allowed values are [auto, diploid, haploid] (Default is auto) |
|
|
Seed for the random number generator. |
|
|
Sex of individual. Allowed values are [male, female, either] (Default is either) |
|
|
If set, display information regarding haplotype selection and crossover points. |
|
Usage:
The childsim command is used to simulate an individuals genotype
information from a VCF containing the two parent genotypes generated by
previous samplesim or childsim commands. The new output VCF will
contain all the existing variants and samples with a new column for the
new sample.
The ploidy for each genotype is generated according to the ploidy of
that chromosome for the specified sex of the individual, as defined in
the reference genome reference.txt file. For more information see
RTG reference file format. The generated genotypes are all consistent
with Mendelian inheritance (de novo variants can be simulated with the
denovosim command).
The --output-sdf flag can be used to optionally generate an SDF of the
child’s genotype which can then be used by the readsim command to
simulate a read set for the child.
By default positions for crossover events are chosen according to a
uniform distribution. However, if linkage information is available,
then this can be used to inform the crossover selection procedure.
The expected format for this information is described in
Genetic map directory, and the directory containing the relevant
files can be specified by using the --genetic-map-dir flag.
See also
pedsamplesim¶
Synopsis:
Generates simulated genotypes for all members of a
pedigree. pedsamplesim automatically simulates founder individuals,
inheritance by children, and de novo mutations.
Syntax:
$ rtg pedsamplesim [OPTION]... -i FILE -o DIR -p FILE -t SDF
Example:
$ rtg pedsamplesim -t reference.sdf -p family.ped -i popvars.vcf \
-o family_sim --remove-unused
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Input VCF containing parent variants. |
|
|
Directory for output. |
|
If set, output an SDF for the genome of each simulated sample. |
|
|
|
Genome relationships PED file. |
|
|
SDF containing the reference genome. |
Utility |
||
|---|---|---|
|
Probability of extra crossovers per chromosome (Default is 0.01) |
|
|
If set, load genetic maps from this directory for recombination point selection. |
|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Expected number of mutations per genome (Default is 70) |
|
|
Ploidy to use. Allowed values are [auto, diploid, haploid] (Default is auto) |
|
|
If set, output only variants used by at least one sample. |
|
|
Seed for the random number generator. |
|
Usage:
The pedsamplesim uses the methods of samplesim, denovosim,
and childsim to greatly ease the simulation of multiple samples. The
input VCF should contain standard allele frequency INFO annotations that
will be used to simulate genotypes for any sample identified as a
founder. Any samples present in the pedigree that are already present in
the input VCF will not be regenerated. To simulate genotypes for a
subset of the members of the pedigree, use pedfilter to create a
filtered pedigree file that includes only the subset required.
The supplied pedigree file is first examined to identify any individuals that cannot be simulated according to inheritance from other samples in the pedigree. Note that simulation according to inheritance requires both parents to be present in the pedigree. These samples in the pedigree are treated as founder individuals.
Founder individuals are simulated using samplesim, where the
genotypes are chosen according to the allele frequency annotation in the
input VCF.
All newly generated samples may have de novo mutations introduced
according to the --num-mutations setting. As with the denovosim
command, any de novo mutations introduced in a sample will be
genotyped as homozygous reference in other pre-existing samples, and
introduced variants will not overlap any pre-existing variant loci.
Samples that can be simulated according to Mendelian inheritance are
then generated, using childsim. As expected, as well as inheriting
de novo variants from parents, each child may obtain new de novo
mutations of their own.
If the simulated samples will be used for subsequent simulated
sequencing, such as via readsim, it is possible to automatically
output an SDF containing the simulated genome for each sample by
specifying the --output-sdf option, obviating the need to separately
use samplereplay.
By default positions for crossover events are chosen according to a
uniform distribution. However, if linkage information is available,
then this can be used to inform the crossover selection procedure.
The expected format for this information is described in
Genetic map directory, and the directory containing the relevant
files can be specified by using the --genetic-map-dir flag.
samplereplay¶
Synopsis:
Use the samplereplay command to generate the genome SDF corresponding
to a sample genotype in a VCF file.
Syntax:
$ rtg samplereplay [OPTION]... -i FILE -o SDF -t SDF -s STRING
Example:
$ rtg samplereplay -i 3samples.vcf -o child.sdf -t HUMAN_reference -s person3
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Input VCF containing the sample genotype. |
|
|
Name for output SDF. |
|
|
SDF containing the reference genome. |
|
|
Name of the sample to select from the VCF. |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
Usage:
The samplereplay command can be used to generate an SDF of a genotype
for a given sample from an existing VCF file. This can be used to
generate a genome from the outputs of the samplesim and childsim
commands. The output genome can then be used in simulating a read set
for the sample using the readsim command.
Every chromosome for which the individual is diploid will have two sequences in the resulting SDF.
Utility Commands¶
bgzip¶
Synopsis:
Block compress a file or decompress a block compressed file. Block
compressed outputs from the mapping and variant detection commands can
be indexed with the index command. They can also be processed with
standard gzip tools such as gunzip and zcat.
Syntax:
$ rtg bgzip [OPTION]... FILE+
Example:
$ rtg bgzip alignments.sam
Parameters:
File Input/Output |
||
|---|---|---|
|
|
The compression level to use, between 1 (least but fast) and 9 (highest but slow) (Default is 5) |
|
|
Decompress. |
|
|
Force overwrite of output file. |
|
If set, do not add the block gzip termination block. |
|
|
|
Write on standard output, keep original files unchanged. Implied when using standard input. |
|
File to (de)compress, use ‘-’ for standard input. Must be specified 1 or more times. |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
Usage:
Use the bgzip command to block compress files. Files such as VCF, BED,
SAM, TSV must be block-compressed before they can be indexed for fast
retrieval of records corresponding to specific genomic regions.
See also
index¶
Synopsis:
Create tabix index files for block compressed TAB-delimited genome position data files or BAM index files for BAM files.
Syntax:
Multi-file input specified from command line:
$ rtg index [OPTION]... FILE+
Multi-file input specified in a text file:
$ rtg index [OPTION]... -I FILE
Example:
$ rtg index -f sam alignments.sam.gz
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Format of input to index. Allowed values are [sam, bam, cram, sv, coveragetsv, bed, vcf, auto] (Default is auto) |
|
|
File containing a list of block compressed files (1 per line) containing genome position data. |
|
Block compressed files containing data to be indexed. May be specified 0 or more times. |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
Usage:
Use the index command to produce tabix indexes for block compressed
genome position data files like SAM files, VCF files, BED files, and the
TSV output from RTG commands such as coverage. The index command
can also be used to produce BAM indexes for BAM files with no index.
extract¶
Synopsis:
Extract specified parts of an indexed block compressed genome position data file.
Syntax:
Extract whole file:
$ rtg extract [OPTION]... FILE
Extract specific regions:
$ rtg extract [OPTION]... FILE STRING+
Example:
$ rtg extract alignments.bam 'chr1:2500000~1000'
Parameters:
File Input/Output |
||
|---|---|---|
|
The indexed block compressed genome position data file to extract. |
|
Filtering |
||
|---|---|---|
|
The range to display. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding>. May be specified 0 or more times. |
|
Reporting |
||
|---|---|---|
|
Set to also display the file header. |
|
|
Set to only display the file header. |
|
Utility |
||
|---|---|---|
|
|
Prints help on command-line flag usage. |
Usage:
Use the extract command to view specific parts of indexed block
compressed genome position data files such as those in SAM/BAM/BED/VCF
format.
aview¶
Synopsis:
View read mapping and variants corresponding to a region of the genome, with output as ASCII to the terminal, or HTML.
Syntax:
$ rtg aview [OPTION]... --region STRING -t SDF FILE+
Example:
$ rtg aview -t hg19 -b omni.vcf -c calls.vcf map/alignments.bam \
--region Chr10:100000+3 –padding 30
Parameters:
File Input/Output |
||
|---|---|---|
|
|
VCF file containing baseline variants. |
|
|
BED file containing regions to overlay. May be specified 0 or more times. |
|
|
VCF file containing called variants. May be specified 0 or more times. |
|
|
File containing a list of SAM/BAM format files (1 per line) |
|
|
Read SDF (only needed to indicate correctness of simulated read mappings). May be specified 0 or more times. |
|
|
SDF containing the reference genome. |
|
Alignment SAM/BAM files. May be specified 0 or more times. |
|
Filtering |
||
|---|---|---|
|
|
Padding around region of interest (Default is to automatically determine padding to avoid read truncation) |
|
The region of interest to display. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
|
Specify name of sample to select. May be specified 0 or more times, or as a comma separated list. |
|
Reporting |
||
|---|---|---|
|
Output as HTML. |
|
|
Do not use base-colors. |
|
|
Do not use colors. |
|
|
Display nucleotide instead of dots. |
|
|
Print alignment cigars. |
|
|
Print alignment MAPQ values. |
|
|
Print mate position. |
|
|
Print read names. |
|
|
Print read group id for each alignment. |
|
|
Print reference line every N lines (Default is 0) |
|
|
Print sample id for each alignment. |
|
|
Print soft clipped bases. |
|
|
If set, project highlighting for the specified track down through reads (Default projects the union of tracks) |
|
|
Sort reads first on read group and then on start position. |
|
|
Sort reads on start position. |
|
|
Sort reads first on sample id and then on start position. |
|
|
Display unflattened CGI reads when present. |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
Usage:
Use the aview command to display a textual view of mappings and
variants corresponding to a small region of the reference genome. This
is useful when examining evidence for variant calls in a server
environment where a graphical display application such as IGV is not
available. The aview command is easy to script in order to output
displays for multiple regions for later viewing (either as text or
HTML).
sdfstats¶
Synopsis:
Print statistics that describe a directory of SDF formatted data.
Syntax:
$ rtg sdfstats [OPTION]... SDF+
Example:
$ rtg sdfstats human_READS_SDF
Location : C:\human_READS_SDF
Parameters : format -f solexa -o human_READS_SDF
c:\users\Elle\human\SRR005490.fastq.gz
SDF Version : 6
Type : DNA
Source : SOLEXA
Paired arm : UNKNOWN
Number of sequences: 4193903
Maximum length : 48
Minimum length : 48
N : 931268
A : 61100096
C : 41452181
G : 45262380
T : 52561419
Total residues : 201307344
Quality scores available on this SDF
Parameters:
File Input/Output |
||
|---|---|---|
|
Specifies an SDF on which statistics are to be reported. May be specified 1 or more times. |
|
Reporting |
||
|---|---|---|
|
Set to print out the name and length of each sequence. (Not recommended for read sets). |
|
|
|
Set to include information about unknown bases (Ns) by read position. |
|
|
Set to display mean of quality. |
|
Set to display the reference sequence list for the given sex. Allowed values are [male, female, either]. May be specified 0 or more times, or as a comma separated list. |
|
|
Set to display information about the taxonomy. |
|
|
|
Set to include information about unknown bases (Ns). |
Utility |
||
|---|---|---|
|
|
Prints help on command-line flag usage. |
Usage:
Use the sdfstats command to get information about the contents of
SDFs.
sdfsubset¶
Synopsis:
Extracts a specified subset of sequences from one SDF and outputs them to another SDF.
Syntax:
Individual specification of sequence ids:
$ rtg sdfsubset [OPTION]... -i SDF -o SDF STRING+
File list specification of sequence ids:
$ rtg sdfsubset [OPTION]... -i SDF -o SDF -I FILE
Example:
$ rtg sdfsubset -i reads -o subset_reads 10 20 30 40 50
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Specifies the input SDF. |
|
|
The name of the output SDF. |
Filtering |
||
|---|---|---|
|
Only output sequences with sequence id less than the given number. (Sequence ids start at 0). |
|
|
Only output sequences with sequence id greater than or equal to the given number. (Sequence ids start at 0). |
|
|
|
Name of a file containing a list of sequences to extract, one per line. |
|
Interpret any specified sequence as names instead of numeric sequence ids. |
|
|
Specifies the sequence id, or sequence name if the names flag is set to extract from the input SDF. May be specified 0 or more times. |
|
Utility |
||
|---|---|---|
|
|
Prints help on command-line flag usage. |
Usage:
Use this command to obtain a subset of sequences from an SDF. Either
specify the subset on the command line as a list of space-separated
sequence ids or using the --id-file parameter to specify a file
containing a list of sequence ids, one per line. Sequence ids start from
zero and are the same as the ids that map uses by default in the
QNAME field of its BAM files.
For example:
$ rtg sdfsubset -i reads -o subset_reads 10 20 30 40 50
This will produce an SDF called subset_reads with sequences 10, 20, 30, 40 and 50 from the original SDF contained in it.
sdfsubseq¶
Synopsis:
Prints a subsequence of a given sequence in an SDF.
Syntax:
Print sequences from sequence names:
$ rtg sdfsubseq [OPTION]... -i FILE STRING+
Print sequences from sequence ids:
$ rtg sdfsubseq [OPTION]... -i FILE -I STRING+
Example:
$ rtg sdfsubseq -i reads -I 0:1+100
Parameters:
File Input/Output |
||
|---|---|---|
|
|
Specifies the input SDF. |
Filtering |
||
|---|---|---|
|
|
If set, use sequence id instead of sequence name in region (0-based) |
|
The range to display. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding>. Must be specified 1 or more times. |
|
Utility |
||
|---|---|---|
|
|
Set to output in FASTA format. |
|
|
Set to output in FASTQ format. |
|
|
Prints help on command-line flag usage. |
|
|
Set to output in reverse complement. |
Usage:
Prints out the nucleotides or amino acids of specified regions in a set of sequences.
For example:
$ rtg sdfsubseq --input reads --sequence-id 0:1+20
AGGCGTCTGCAGCCGACGCG
mendelian¶
Synopsis:
The mendelian command checks a multi-sample VCF file for variant calls
which do not follow Mendelian inheritance, and compute aggregate sample
concordance.
Syntax:
$ rtg mendelian [OPTION]... -i FILE -t SDF
Example:
$ rtg mendelian -i family.vcf.gz -t genome_ref
Parameters:
File Input/Output |
||
|---|---|---|
|
|
VCF file containing multi-sample variant calls. Use ‘-’ to read from standard input. |
|
|
If set, output annotated calls to this VCF file. Use ‘-’ to write to standard output. |
|
If set, output only consistent calls to this VCF file. |
|
|
If set, output only non-Mendelian calls to this VCF file. |
|
|
|
SDF containing the reference genome. |
Sensitivity Tuning |
||
|---|---|---|
|
Use all records, regardless of filters (Default is to only process records where FILTER is |
|
|
|
Allow homozygous diploid calls in place of haploid calls and assume missing values are equal to the reference. |
|
Percentage concordance required for consistent parentage (Default is 99.0) |
|
|
Genome relationships PED file (Default is to extract pedigree information from VCF header fields) |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
Usage:
Given a multi-sample VCF file for a nuclear family with a defined
pedigree, the mendelian command examines the variant calls and outputs
the number of violations of Mendelian inheritance. If the
--output-inconsistent parameter is set, all detected violations are
written into an output VCF file. As such, this command may be regarded
as a VCF filter, outputting those variant calls needing a non-Mendelian
explanation. Such calls may be the consequence of sequencing error,
calling on low-coverage, or genuine novel variants in one or more
individuals.
Pedigree information regarding the relationships between samples and the
sex of each sample is extracted from the VCF headers automatically
created by the RTG pedigree-aware variant calling commands. If this
pedigree information is absent from the VCF header or is incorrect, a
pedigree file can be explicitly supplied with the --pedigree flag.
To ensure correct behavior when dealing with sex chromosomes it is
necessary to specify a sex-aware reference and ensure the sex of each
sample is supplied as part of the pedigree information. While it is best
to give the reference SDF used in the creation of the VCF, for checking
third-party outputs any reference SDF containing the same chromosome
names and an appropriate reference.txt file will work. For more
information, see RTG reference file format. Variants calls where
the call ploidy does not match what is expected are annotated in the
output VCF with an MCP FORMAT annotation.
Particularly when evaluating VCF files that have been produced by third
party tools or when the VCF is the result of combining independent
per-sample calling, it is common to end up with situations where calls
are not available for every member of the family. Under normal
circumstances mendelian will attempt to determine Mendelian
consistency on the basis of the values that have been provided. Records
where the presence of missing values makes the Mendelian consistency
undecidable contain MCU INFO annotations in the annotated output
VCF. The following examples illustrate some consistent, undecidable, and
inconsistent calls in the presence of missing values:
CHROM FATHER_GT MOTHER_GT SON_GT STATUS
chrX . 0/1 1 OK
chr1 ./. 1/1 1/2 MCU
chr1 ./. 1/1 2/2 MCV
Since the number of calls where one sample is missing can be quite high,
an alternative option is to treat missing values as equal to the
reference by using the --lenient parameter. Note that while this
approach will be correct in most cases, it will give inaccurate results
where the calling between different samples has reported the variant in
an equivalent but slightly different position or representation
(e.g. positioning of indels within homopolymer regions, differences of
representation such as splitting MNPs into multiple SNPs etc).
The mendelian command computes overall concordance between related
samples to assist detecting cases where pedigree has been incorrectly
recorded or samples have been mislabeled. For each child in the
pedigree, pairwise concordance is computed with respect to each parent
by identifying diploid calls where the parent does not contain either
allele called in the child. Low pairwise concordance with a single
parent may indicate that the parent is the source of the problem,
whereas low pairwise concordance with both parents may indicate that the
child is the source of the problem. A stricter three-way concordance is
also recorded.
By default, only VCF records with the FILTER field set to PASS or
missing are processed. All variant records can be examined by specifying
the --all-records parameter.
See also
vcfannotate¶
Synopsis:
Used to add annotations to a VCF file, either to the VCF ID field,
as a VCF INFO sub-field, or as a VCF FORMAT sub-field.
Syntax:
$ rtg vcfannotate [OPTION]... -b FILE -i FILE -o FILE
Example:
$ rtg vcfannotate -b dbsnp.bed -i snps.vcf.gz -o snps-dbsnp.vcf.gz
Parameters:
File Input/Output |
||
|---|---|---|
|
If set, only read VCF records that overlap the ranges contained in the specified BED file. |
|
|
|
VCF file containing variants to annotate. Use ‘-’ to read from standard input. |
|
|
Output VCF file name. Use ‘-’ to write to standard output. |
|
If set, only read VCF records within the specified range. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
Reporting |
||
|---|---|---|
|
|
Add computed annotation to VCF records. Allowed values are [AC, AN, EP, GQD, IC, LAL, MEANQAD, NAA, PD, QA, QD, RA, SCONT, VAF, VAF1, ZY]. May be specified 0 or more times, or as a comma separated list. |
|
Add variant IDs from BED file. May be specified 0 or more times. |
|
|
Add INFO annotations from BED file. May be specified 0 or more times. |
|
|
Add or update the AN and AC INFO fields. |
|
|
If the BED INFO field is not already declared, use this description in the header (Default is Annotation) |
|
|
The INFO ID for BED INFO annotations (Default is ANN) |
|
|
Relabel samples according to |
|
|
Add variant IDs from VCF file. May be specified 0 or more times. |
|
Utility |
||
|---|---|---|
|
|
File containing VCF header lines to add, or a literal header line. May be specified 0 or more times. |
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Prevent VCF header from being written. |
|
Usage:
Use vcfannotate to add text annotations to variants.
A common use case is to add annotations to only those variants that fall
within ranges specified in a BED or VCF file, supplied via --bed-ids
or --vcf-ids respectively. The annotations from the BED file are
added as an INFO field in the output VCF file. It can also be used
to compute or fill in certain additional annotations from the existing
content. Note that this annotation method is solely based on the
position and span of the variant, ignoring actual alleles and genotypes.
If the --bed-ids flag is used, instead of adding the annotation to the
INFO fields, it is added to the ID column of the VCF file instead.
If the --vcf-ids flag is used, the ID column of the input VCF file
is used to update the ID column of the output VCF file instead.
If the --fill-an-ac flag is set, the output VCF will have the AN and
AC info fields (as defined in the VCF 4.2 specification) created or
updated.
It is also possible to use vcfannotate to insert additional VCF
header lines into the VCF header. These are supplied using the
--add-header flag which may either be a literal VCF header line
(useful for adding one or two header lines), or from a file.
$ rtg vcfannotate -i in.vcf.gz -o out.vcf.gz \
--add-header "##SAMPLE=<ID=NA24385,Sex=MALE>" \
--add-header "##SAMPLE=<ID=NA24143,Sex=FEMALE>" \
--add-header "##SAMPLE=<ID=NA24149,Sex=MALE>" \
--add-header "##PEDIGREE=<Child=NA24385,Mother=NA24143,Father=NA24149>"
or alternatively:
$ rtg vcfannotate -i in.vcf.gz -o out.vcf.gz --add-header ped_vcf_headers.txt
Care should be taken that the lines being inserted are valid VCF header lines.
If the --annotation flag is set, vcfannotate attempts to compute
the specified annotation(s) and add them as FORMAT fields in the
corresponding records. Records for which particular annotations cannot be
computed, due to a lack of pre-requisite fields, will not be modified.
For a description of the meaning of fields available for annotation,
see Small-variant VCF output file description. The SCONT
annotation is a convenience to annotate with all of the contrary
evidence annotations: DCOC, DCOF, OCOC, OCOF.
vcfdecompose¶
Synopsis:
Decomposes complex variants within a VCF file into smaller components.
Syntax:
$ rtg vcfdecompose [OPTION]... -i FILE -o FILE
Parameters:
File Input/Output |
||
|---|---|---|
|
|
VCF file containing variants to decompose. Use ‘-’ to read from standard input. |
|
|
Output VCF file name. Use ‘-’ to write to standard output. |
|
|
SDF of the reference genome the variants are called against. |
Sensitivity Tuning |
||
|---|---|---|
|
If set, peel as many SNPs off an indel as possible. |
|
|
If set, break MNPs into individual SNPs. |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Prevent VCF header from being written. |
|
Usage:
The vcfdecompose command decomposes and trims variants based on
a multiple sequence alignment between the alleles in each VCF
record. Only records where every ALT allele is an ordinary allele
(i.e. consisting of nucleotides) will undergo decomposition. In
addition, if there are redundant same-as-reference bases in the alleles,
these will be trimmed off.
The default behavior is to break the variant at positions where there
is at least one base aligned to the reference across all ALT alleles, so
the output may contain MNPs or impure indels. If desired, MNPs can be
split into individual SNPs via --break-mnps. Similarly, impure
indels can be split into a combination of SNPs and pure indels via
--break-indels.
Although decomposed variants carry through the original INFO and
FORMAT annotations, the decomposition may mean that some annotations
are no longer semantically correct. In particular, any VCF FORMAT
fields declared to be of type A, G, or R will no longer be
valid if the set of alleles has changed.
Note that the reference genome is an optional parameter. When variants
are decomposed and trimmed, the resulting variant may require a padding
base to be added, as required by the VCF specification. The VCF
specification suggests that the padding base should be the base before
the variant (i.e. padding on the left), but sometimes this requires
knowledge of reference bases not present in the original record. When
the reference genome is supplied, vcfdecompose will ensure that any
padding bases are added on the left of the variant. If the reference
genome is not supplied, padding bases may sometimes be on the right hand
side of the variant. For example:
1 20 . GCGCGCGCGCG TTTGCGCGCTTGCGCGTTT . PASS . GT 1/0
will decompose without a reference genome as:
1 20 . G TTTG . PASS ORP=20;ORL=11 GT 1/0
1 25 . C CTT . PASS ORP=20;ORL=11 GT 1/0
and with a reference genome (where the reference base at position 19 can
be determined to be a T) as:
1 19 . T TTTT . PASS ORP=20;ORL=11 GT 1/0
1 25 . C CTT . PASS ORP=20;ORL=11 GT 1/0
The variants that are left vs right-padded are equivalent and identified
as such by haplotype-aware comparison tools such as vcfeval.
vcfeval¶
Synopsis:
Evaluates called variants for agreement with a baseline variant set
irrespective of representational differences. Outputs a weighted ROC
file which can be viewed with rtg rocplot and VCF files containing false
positives (called variants not matched in the baseline), false negatives
(baseline variants not matched in the call set), and true positives
(variants that match between the baseline and calls).
The baseline variants might be the variants that were used to generate a
synthetic simulated sample (such as via popsim, samplesim, etc),
a gold-standard VCF corresponding to a reference sample such as NA12878,
or simply an alternative call-set being used as a basis for comparison.
Syntax:
$ rtg vcfeval [OPTION]... -b FILE -c FILE -o DIR -t SDF
Example:
$ rtg vcfeval -b goldstandard.vcf.gz -c snps.vcf.gz -t HUMAN_reference \
--sample daughter -f AVR -o eval
Parameters:
File Input/Output |
||
|---|---|---|
|
|
VCF file containing baseline variants. |
|
If set, only read VCF records that overlap the ranges contained in the specified BED file. |
|
|
|
VCF file containing called variants. |
|
|
If set, evaluate within regions contained in the supplied BED file, allowing transborder matches. To be used for truth-set high-confidence regions or other regions of interest where region boundary effects should be minimized. |
|
|
Directory for output. |
|
If set, only read VCF records within the specified range. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
|
|
SDF of the reference genome the variants are called against. |
Filtering |
||
|---|---|---|
|
Use all records regardless of filter status (Default is to only process variants passing filters) |
|
|
Decompose complex variants into smaller constituents to allow partial credit. |
|
|
Allow alleles to overlap where bases of either allele are same-as-ref (Default is to only allow VCF anchor base overlap) |
|
|
The name of the sample to select. Use <baseline_sample>,<calls_sample> to select different sample names for baseline and calls. (Required when using multi-sample VCF files) |
|
|
Expected ploidy of samples (Default is 2) |
|
|
Treat heterozygous genotypes as homozygous ALT in both baseline and calls, to allow matches that ignore zygosity differences. |
|
Reporting |
||
|---|---|---|
|
Output summary statistics where precision >= supplied value (Default is to summarize at maximum F-measure) |
|
|
Output summary statistics where score meets supplied value (Default is to summarize at maximum F-measure) |
|
|
Output summary statistics where sensitivity >= supplied value (Default is to summarize at maximum F-measure) |
|
|
Do not produce ROCs. |
|
|
|
Output reporting mode. Allowed values are [split, annotate, combine, ga4gh, roc-only] (Default is split) |
|
Output ROC files for each intersection of –roc-subset, –roc-expr, and –roc-regions values provided. |
|
|
Output ROC file for variants matching custom JavaScript expression. Use the form <LABEL>=<EXPRESSION>. May be specified 0 or more times. |
|
|
Output ROC file for variants overlapping custom regions supplied in BED file. Use the form <LABEL>=<FILENAME>. May be specified 0 or more times. |
|
|
Output ROC file for preset variant subset. Allowed values are [hom, het, snp, non-snp, mnp, indel]. May be specified 0 or more times, or as a comma separated list. |
|
|
|
The order in which to sort the ROC scores so that |
|
|
The name of the VCF FORMAT field to use as the ROC score. Also valid are |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
|
Number of threads (Default is the number of available cores) |
Usage:
The vcfeval command can be used to generate VCF files containing
called variants that were in the baseline VCF, called variants that were
not in the baseline VCF and baseline variants that were not in the
called variants. It also produces ROC curve data files based on a score
contained in a VCF field which show the predictive power of that field
for the quality of the variant calls.
When developing and validating sequencing pipelines and variant calling
algorithms, the comparison of variant call sets is a common problem. The
naïve way of computing these numbers is to look at the same reference
locations in the baseline (ground truth) and called variant set, and see
if genotype calls match at the same position. However, a complication
arises due to possible differences in representation for indels between
the baseline and the call sets within repeats or homopolymers, and in
multiple-nucleotide polymorphisms (MNPs), which encompass several nearby
nucleotides and are locally phased. The vcfeval command includes a
novel dynamic-programming algorithm for comparing variant call sets that
deals with complex call representation discrepancies, and minimizes
false positives and negatives across the entire call sets for accurate
performance evaluation. A primary advantage of vcfeval (compared to
other tools) is that the evaluation does not depend on normalization or
decomposition, and so the results of analysis can easily be used to
relate to the original variant calls and their annotations.
Note that vcfeval operates at the level of local haplotypes for a
sample, so for a diploid genotype, both alleles must match in order to
be considered correct. Some of the vcfeval output modes (described
below) automatically perform an additional haploid analysis phase to
identify variants which may not have a diploid match but which share a
common allele (for example, zygosity errors made during calling). If
desired, this more lenient haploid comparison can be used at the outset
by setting the --squash-ploidy flag (see below).
A few categories of variants are excluded from the matching process:
Those variants that have failed filters, according to the record
FILTERfield, or the per-sampleFTFORMATfield (when applicable), unless--all-recordsis specified.Variants that are not resolved at the nucleotide level, such as CNVs and structural variants.
Variants that do not represent variation from the reference (for example reference calls).
Variants where
REForALTexceeds a maximum length of 1000 nucleotides, for performance reasons.Variants which are not fully enclosed by the relevant reference sequence.
Variants which do not overlap the regions specified via
--bed-regions, when applicable.
Variants selected for inclusion in a haplotype cannot be
permitted to overlap each other (otherwise the question arises of which
variant should have priority when determining the resulting haplotype),
and any well-formed call-set should not contain these situations in
order to avoid such ambiguity. When such cases are encountered by
vcfeval, the best non-overlapping result is determined. A special case
of overlapping variants is where calls are denoted as partially the same
as the reference (for example, a typical heterozygous call). Strictly
speaking such variants are an assertion that the relevant haplotype
bases must not be altered from the reference and overlap should not be
permitted (this is the interpretation that vcfeval employs by
default). However, sometimes as a result of using non-haplotype-aware
variant calling tools or when using naïve merging of multiple call sets,
a more lenient comparison is desired. The --ref-overlap flag will
permit such overlapping variants to both match, as long as any overlap
only occurs where one variant or other has asserted haplotype bases as
being the same as reference.
Common allele matching with --squash-ploidy¶
When --squash-ploidy is specified, a haploid match is attempted
using each of the non-reference alleles used in the sample
genotype. For example if the baseline and call VCFs each had a record
with the same REF and ALT alleles declared, the following GT
fields would be considered a match:
0/1, 1/1, 1/2 (genotypes match due to the 1 allele)
0/2, 1/2, 2/2 (genotypes match due to the 2 allele)
Thus --squash-ploidy matches any case where the baseline and calls
share a common allele. This is most often used to run matching that
does not penalize for genotyping errors. For example, it is recommended
to use this option when matching somatic variant calls, as since somatic
variation is usually associated with variable allelic fractions and
heterogeneity that mean strict diploid genotype comparisons are not
appropriate.
Comparing non-diploid genomes¶
By default, vcfeval assumes diploid organisms (that is, the expected
ploidy of any GT call is 2). As a special case to ease the
comparison of male calls on sex chromosomes (where callers often
continue to use diploid representation), haploid calls are treated as
homozygous diploid. Any calls made with unexpected ploidy are ignored
and reported in the vcfeval log file.
To compare genomes with non-diploid ploidy, the expected sample ploidy
can be overridden via --sample-ploidy – for example
--sample-ploidy=4 would be used to compare tetraploid organisms.
Comparing with a VCF that has no sample column¶
A common scenario is to match a call set against a baseline which
contains no sample column, where the objective is to identify which
baseline alleles which have been called. One example of this is to
identify whether calls match a database of known high-priority somatic
variants such as COSMIC, or to find calls which have been previously
seen in a population allele database such as ExAC. Ordinarily
vcfeval requires the input VCFs to contain a sample column
containing a genotype in the GT field, however, it is possible to
specify a special sample name of ‘ALT’ in order to indicate that the the
genotypes for comparison should be derived from the ALT alleles of
the record. This can be specified independently for baseline and calls,
for example:
$ rtg vcfeval -t build37.sdf -b cosmic.vcf.gz -c tumor-calls.vcf.gz \
--squash-ploidy --sample ALT,tumor -o tumor-vs-cosmic
Which would perform a haploid matching of the GT of the called
sample ‘tumor’ against all possible haploid genotypes in the COSMIC
VCF. The resulting true positives file contains all the calls containing
an allele present in the COSMIC VCF.
Note
It is also possible to run a diploid comparison by omitting
--squash-ploidy, but this is not usually required, and is
computationally more intensive since there may be many more possible
diploid genotypes to explore, particularly if the ALT VCF contains
many multi-allelic records.)
Evaluation with respect to regions¶
When evaluating exome variant calls, it may be useful to restrict
analysis only to exome target regions. In this case, supply a BED file
containing the list of regions to restrict analysis to via the
--bed-regions flag. For a quick way to restrict analysis only to a
single region, the --region flag is also accepted. Note that when
restricting analysis to regions, there may be variants which can not be
correctly evaluated near the borders of each analysis region, if
determination of equivalence would require inclusion of variants outside
of the region. For this reason, it is recommended that such regions be
relatively inclusive.
When matching against gold standard truth sets which have an
accompanying high-confidence regions BED file, the flag
--evaluation-regions should be used instead of --bed-regions, as
it has special matching semantics that aims to reduce comparison region
boundary effects. When this comparison method is used, call variants
which match a baseline variant are only considered a true positive if
the baseline variant is inside the high confidence regions, and call
variants are only considered false positive if they fall inside the high
confidence regions.
vcfeval outputs¶
The primary outputs of vcfeval are VCF files indicating which variants
matched between the baseline and the calls VCF, and data files
containing information used to generate ROC curves with the rocplot
command (or via spreadsheet). vcfeval supports different VCF
output modes which can be selected with the --output-mode flag
according to the type of analysis workflow desired. The following modes
are available:
Split (--output-mode=split)¶
This output mode is the default, and produces separate VCF files for each of the match categories. The individual VCF records in these files are not altered in any way, preserving all annotations present in the input files.
tp.vcf– contains those variants from the calls VCF which agree with variants in the baseline VCFtp-baseline.vcf– contains those variants from the baseline VCF which agree with variants in the calls VCF. Thus, the variants intp.vcfandtp-baseline.vcfare equivalent. This file can be used to successively refine a highly sensitive baseline variant set to produce a consensus from several call sets.fp.vcf– contains variants from the calls VCF which do not agree with baseline variants.fn.vcf– contains variants from the baseline VCF which were not correctly called.
This mode performs a single pass comparison, either in diploid mode (the
default), or haploid mode (if --squash-ploidy has been set). The
separate output files produced by this mode allow the use of vcfeval
as an advanced haplotype-aware VCF intersection tool.
Annotate (--output-mode=annotate)¶
This output mode does not split the input VCFs by match status, but
instead adds INFO annotations containing the match status of each
record:
calls.vcf– contains variants from the calls VCF, augmented with match status annotations.baseline.vcf– contains variants from the baseline VCF, augmented with match status annotations.
This output mode automatically performs two comparison passes, the first
finds diploid matches (assigned a match status of TP), and a second
pass that applies a haploid mode to the false positives and false
negatives in order to find calls (such as zygosity errors) that contain
a common allele. This second category of match are annotated with status
FN_CA or FP_CA in the output VCFs, and those calls which do not
have any match are assigned status FN or FP. A status value of
IGN indicates a VCF record which was ignored (for example, due to
having a non-PASS filter status, representing a structural variant, or
otherwise containing a non-variant genotype). A status of OUT
indicates a VCF record which does not contain a match status due to
falling outside the evaluation regions when --evaluation-regions is
being used. The annotated VCF files produced in this mode may also be
used with vcf2rocplot to produce additional post-evaluation ROC data
files.
Combine (--output-mode=combine)¶
This output mode provides an easy way to view the baseline and call variants in a single two-sample VCF.
output.vcf– contains variants from both the baseline and calls VCFs, augmented with match status annotations. The sample under comparison from each of the input VCFs is extracted as a column in the output. As the VCF records from the baseline and calls typically have very different input annotations which can be difficult to merge, and to keep the output format simple, there is no attempt to preserve any of the original variant annotations.
As with the annotation output mode, this output mode automatically performs two comparison passes to find both diploid matches and haploid (lenient) matches.
ROC-only (--output-mode=roc-only)¶
This output mode provides a lightweight way to run performance benchmarking, as VCF file output is omitted, and only ROC data files are produced.
Note
In addition, vcfeval has an output mode
(--output-mode=ga4gh) which produces the intermediate evaluation
format defined by the GA4GH Benchmarking Team, without additional
statistics files. This mode is not generally intended for end users,
rather it is used when vcfeval is selected as the comparison engine
inside the hap.py benchmarking tool see:
https://github.com/ga4gh/benchmarking-tools and
https://github.com/Illumina/hap.py
Additional ROC stratifications¶
All of the output modes produce the following ROC data files (unless
disabled by --no-roc):
weighted_roc.tsv– contains ROC data derived from all analyzed call variants, regardless of their representation. Columns include the score field, and standard accuracy metrics such as true positives, false positives, false negatives, precision, sensitivity, and f-measure corresponding to each score threshold.snp_roc.tsv– contains ROC data derived from only those variants which were represented as SNPs. Since the representation conventions can differ between the baseline and calls, there are some subtleties to be aware of when interpreting metrics such as precision, sensitivity, etc, described below.non_snp_roc.tsv– contains ROC data derived from those variants which were not represented as SNPs. As above, not all metrics are computed for this file.
vcfeval also provides the ability to produce additional ROC data
files corresponding to preset and customized variant stratifications
with the following flags:
Preset stratifications¶
The --roc-subset flag allows selection from preset stratifications.
The following presets are based on variant type (according to their representation in the relevant input VCF):
snp– SNP variants (enabled by default)non-snp– non-SNP variants (enabled by default)mnp– multi-nucleotide polymorphisms onlyindel– length-changing variants only
The following preset stratifications are based on the variant genotype:
hom– homozygous variants onlyhet– heterozygous variants only
Multiple presets can be enabled in a single run, e.g. --roc-subset
hom,het,indel. See below for more information regarding multiple
stratifications.
Region-based stratifications¶
The --roc-regions flag produces a stratified ROC data file using only
variants that overlap regions specified in a user-supplied BED file. The
special syntax for this flag is: --roc-regions LABEL=FILE, where
LABEL is a short tag used to determine ROC output file names and
FILE is the path to the relevant BED file. For example, to produce
additional stratifications based on BED files partitioning the genome
based on GC content:
$ rtg vcfeval -t build37.sdf -b baseline.vcf.gz -c calls.vcf.gz \
--roc-regions GC55TO60=/path/to/GCcontent/GRCh37_gc55to60_slop50.bed.gz \
--roc-regions GC60TO65=/path/to/GCcontent/GRCh37_gc60to65_slop50.bed.gz
Custom JavaScript based stratifications¶
The above stratification flags will satisfy most common usages, but
vcfeval also includes the ability to write custom stratifications
using JavaScript expressions (similar to vcffilter --keep-expr). The
special syntax for this flag is: --roc-expr LABEL=EXPRESSION, where
LABEL is a short tag used to determine ROC output file names and
EXPRESSION is the JavaScript expression that accepts a variant for
inclusion in the stratification. This is most useful when the input VCFs
contain annotations useful for the stratification. For example, to
produce stratifications based on depth of coverage during variant
calling:
$ rtg vcfeval -t build37.sdf -b baseline.vcf.gz -c calls.vcf.gz \
--roc-expr "DP10TO20=has(INFO.DP) && INFO.DP>=10 && INFO.DP<20" \
--roc-expr "DP20TO30=has(INFO.DP) && INFO.DP>=20 && INFO.DP<30" \
--roc-expr "DP30TO40=has(INFO.DP) && INFO.DP>=30 && INFO.DP<40"
Tips:
Ensure the expression is valid to evaluate on all variants (for example, take care when referring to sample fields names if the sample names are different between baseline and calls files).
It may be useful to test or debug the expression (without the label) via
vcffilter --keep-expr.
For more information on JavaScript expressions, see RTG JavaScript filtering API
Intersecting multiple stratifications¶
The default behaviour of vcfeval is to treat each stratification independently
and output one ROC file for each selected stratification. In some cases it is
useful to produce an output file for each possible intersection of the
selected ROC categories. For example, to generate separate ROC curves for SNPs
and non-SNPs within each of multiple specified BED region files. This can be
accomplished with the --roc-cross-join flag, for example:
$ rtg vcfeval -t build37.sdf -b baseline.vcf.gz -c calls.vcf.gz \
--roc-cross-join \
--roc-subset snp,non-snp \
--roc-regions GC55TO60=/path/to/GCcontent/GRCh37_gc55to60_slop50.bed.gz \
--roc-regions GC60TO65=/path/to/GCcontent/GRCh37_gc60to65_slop50.bed.gz
This will produce the four ROC output files: GC55TO60+snp_roc.tsv.gz,
GC55TO60+non_snp_roc.tsv.gz, GC60TO65+snp_roc.tsv.gz, and
GC60TO65+non_snp_roc.tsv.gz.
Benchmarking comparisons using ROC and precision/sensitivity curves¶
Multiple ROC data files (from a single or several vcfeval runs) can
be plotted with the rocplot command, which allows output to a PNG or
SVG image or analysis in an interactive GUI that provides zooming and
visualization of the effects of threshold adjustment. As these files are
simple tab-separated-value format, they can also be loaded into a
spreadsheet tool or processed with shell scripts.
While ROC curve analysis provides a much more thorough method for
examining the performance of a call set with respect to a baseline truth
set, for convenience, vcfeval also produces a summary.txt file
which indicates match summary statistics that correspond to two key
points on the ROC curve. The first point is where all called variants
are included (i.e., no thresholding on a score value); and second point
corresponding to a score threshold that maximises the F-measure of the
curve. While this latter point is somewhat arbitrary, it represents a
balanced tradeoff between precision and sensitivity which is likely to
provide a fairer comparison when comparing call sets from different
callers.
Sometimes it is useful to report the summary statistic evaluation at
some point other than maximized F-measure (for example when comparing a
large number of results at a particular precision level). This can be
accomplished by specifying a different point using either the
--at-precision or --at-sensitivity flag with a value in the
range [0, 1]. Alternatively, you can instruct the summary table to
select the point corresponding to a fixed score threshold of interest.
For example, when using QUAL as a score field, --at-score 20
would list the accuracy statistics equivalent to selecting VCF records
with a QUAL score greater or equal to 20.
Note that vcfeval reports true positives both counted using the
baseline variant representation as well as counted using the call
variant representation. When these numbers differ greatly, it indicates
a general difference in representational conventions used between the
two call sets. Since false negatives can only be measured in terms of
the baseline representation, sensitivity is defined as:

Conversely since false positives can only be measured in terms of the call representation, precision is defined as:
Note
For definitions of the terminology used when evaluating caller accuracy, see: https://en.wikipedia.org/wiki/Receiver_operating_characteristic and https://en.wikipedia.org/wiki/Sensitivity_and_specificity
Benchmarking performance for SNPs versus indels¶
A common desire is to perform analysis separately for SNPs versus indels. However, it is important to note that due the representation ambiguity problem, it is not always trivial to decide in a global sense whether a variant is a SNP or an indel or other complex variant. A group of variants that may be represented as single SNPs in one call-set may be represented as a single complex variant in another call-set. Consider the following example reference and alternate haplotypes:
12345678901234567
REF: ATCGTAAATAAAATGCA
ALT: ATCGTAAAATAAATGCA
One variant caller might represent the haplotypes as the following VCF records:
chr1 5 . T TA . . . GT 1/1
chr1 9 . TA T . . . GT 1/1
While another variant caller could represent the same haplotypes as:
chr1 9 . T A . . . GT 1/1
chr1 10 . A T . . . GT 1/1
The decision as to which representation to use is essentially arbitrary, yet one caller has used indels (and no SNPs), and the other has used SNPs (and no indels). For this reason it is certainly a poor idea to attempt to divide baseline and called variants into separate SNP and indel datasets up front and perform evaluation on each set separately, as any variants that use different representation categories will not be matched across the independent comparisons. Any variant-type specific metrics should be computed after matching is carried out on the full variant sets.
Note that when there are different representational conventions between the baseline and calls (or between calls from one variant caller and another), then at some level there is really a semantic difference between a “baseline indel” and a “call-set indel” (or “variant-caller-A indel” and “variant-caller-B indel”), so caution should be applied when making conclusions related to SNP versus indel accuracy.
In the snp_roc.tsv and non_snp_roc.tsv output files (and other
preset stratifications available via --roc-subset), vcfeval
notes the number of baseline and call variants of each variant
type. When considering benchmarking metrics in the absence of any
thresholding with respect to a score field, it is straight-forward to
use the previous formulae (i.e. sensitivity is computed using the counts
from baseline variants, and precision is computed using the counts from
called variants). When computing threshold-specific metrics for ROC data
points, the computation is more involved. Since only the call variants
contain the score field used to rank variants, the number of (say) TP
baseline indels that exceed threshold  is not
defined.
is not
defined. vcfeval computes a scaled count as:
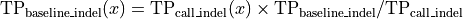
and thus threshold-specific sensitivity is computed as
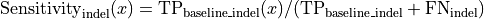
This scaling ensures that the end point of the variant type specific ROC or precision/sensitivity curve ends at the same point that is obtained when computing metrics without any threshold.
The scaling described above is applied to all of the preset
stratifications available via --roc-subset, but is not applied to
any custom stratifications produced via --roc-regions or
--roc-expr.
Variant decomposition and benchmarking¶
In general, it is not necessary to run any variant decomposition and/or
normalization on variant call sets prior to evaluation with vcfeval,
as the haplotype aware matching process can account for representation
differences. However, since matching is at the granularity of entire
variants, a single long complex call will be categorized as either
correct or incorrect, even if part of the call may match. If partial
credit in the case of long calls is of interest, vcfeval includes an
option to internally decompose variants prior to matching, using the
--decompose flag. This decomposition is applied to both baseline and
call variants, and any output VCFs will contain the decomposed
representation. External VCF decomposition (with more control over
decomposition options) is also available via rtg vcfdecompose.
See also
snp, popsim, samplesim, childsim, rocplot, vcf2rocplot, vcfdecompose, bndeval, cnveval
vcffilter¶
Synopsis:
Filters VCF records based on various criteria. When filtering on multiple samples, if any of the specified samples fail the criteria, the record will be filtered. By default filtered records are removed, but see the –fail, –clear-failed-samples, and –fail-samples options for alternatives.
Syntax:
$ rtg vcffilter [OPTION]... -i FILE -o FILE
Examples:
Keep only records where the sample has depth of coverage at least 5:
$ rtg vcffilter -i snps.vcf.gz -o snps_cov5.vcf.gz -d 5
Keep only biallelic records:
$ rtg vcffilter -i snps.vcf.gz -o snps_biallelic.vcf.gz --max-alleles 2
Parameters:
File Input/Output |
||
|---|---|---|
|
Apply sample-specific criteria to all samples contained in the input VCF. |
|
|
If set, only read VCF records that overlap the ranges contained in the specified BED file. |
|
|
|
VCF file containing variants to be filtered. Use ‘-’ to read from standard input. |
|
|
Output VCF file. Use ‘-’ to write to standard output. This option is required, unless |
|
If set, only read VCF records within the specified range. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
|
Apply sample-specific criteria to the named sample contained in the input VCF. May be specified 0 or more times. |
|
Filtering (Record based) |
||
|---|---|---|
|
|
Window within which multiple variants are discarded. |
|
Discard all variants within the regions in this BED file. |
|
|
Discard all variants that overlap with the ones in this file. |
|
|
Only keep variants within the regions in this BED file. |
|
|
Only keep variants that overlap with the ones in this file. |
|
|
|
Javascript filtering functions for determining whether to keep record. May be either an expression or a file name. May be specified 0 or more times. See Examples |
|
|
Records for which this expression evaluates to true will be retained. See Examples |
|
|
Only keep variants with this FILTER tag. May be specified 0 or more times, or as a comma separated list. |
|
|
Only keep variants with this INFO tag. May be specified 0 or more times, or as a comma separated list. |
|
Maximum number of alleles (REF included) |
|
|
|
Maximum allowed combined read depth. |
|
|
Maximum allowed quality. |
|
Minimum number of alleles (REF included) |
|
|
|
Minimum allowed combined read depth. |
|
|
Minimum allowed quality. |
|
|
Remove variants with this FILTER tag. May be specified 0 or more times, or as a comma separated list. |
|
|
Remove variants with this INFO tag. May be specified 0 or more times, or as a comma separated list. |
|
Remove records that overlap with previous records. |
|
Filtering (Sample based) |
||
|---|---|---|
|
|
Maximum allowed ambiguity ratio. |
|
Maximum allowed AVR score. |
|
|
Maximum de novo score threshold. |
|
|
|
Maximum allowed genotype quality. |
|
|
Maximum allowed sample read depth. |
|
Minimum allowed AVR score. |
|
|
Minimum de novo score threshold. |
|
|
|
Minimum allowed genotype quality. |
|
|
Minimum allowed sample read depth. |
|
Only keep where sample variant is MNP or INDEL. |
|
|
Remove where all samples are same as reference. |
|
|
Remove where sample is homozygous. |
|
|
Remove where sample is same as reference. |
|
|
Only keep where sample variant is a simple SNP. |
|
Reporting |
||
|---|---|---|
|
Retain failed records, set the sample GT field to missing. |
|
|
|
Retain failed records, add the provided label to the FILTER field. |
|
|
Retain failed records, add the provided label to the sample FT field. |
Utility |
||
|---|---|---|
|
|
File containing VCF header lines to add, or a literal header line. May be specified 0 or more times. |
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Prevent VCF header from being written. |
|
Usage:
Use vcffilter to get a subset of the results from variant calling
based on the filtering criteria supplied by the filter flags. Multiple
criteria can be specified at once, and advanced processing can be
specified via JavaScript scripting.
When filtering on multiple samples, if any of the specified samples fail
the criteria, the record will be filtered. The default behavior is for
filtered records to be excluded from output altogether, but
alternatively the records can be retained but with an additional
user-specified VCF FILTER status set via --fail option, or if
sample-specific filtering criteria is being applied, only those samples
can be filtered either by setting their GT field to missing by using
the --clear-failed-samples option, or by setting the FORMAT
FT field with a user-specified status via the --fail-samples
option.
The --bed-regions option makes use of tabix indexes to avoid loading
VCF records outside the supplied regions, which can give faster
filtering performance. If the input VCF is not indexed or being read
from standard input, or if records failing filters are to be annotated
via the --fail option, use the --include-bed option instead.
The flags --min-denovo-score and --max-denovo-score can only be used
on a single sample. Records will only be kept if the specified sample is
flagged as a de novo variant and the score is within the range
specified by the flags. It will also only be kept if none of the other
samples for the record are also flagged as a de novo variant within
the specified score range.
The --add-header option allows inserting arbitrary VCF header lines
into the output VCF. For more information, see vcfannotate.
A powerful general-purpose filtering capability has been included that
permits the specification of filter criteria as simple JavaScript
expressions (--keep-expr) or more comprehensive JavaScript processing
functions (--javascript). Both --keep-expr and --javascript can
take JavaScript on the command line or if a filename is supplied then
the script/expression will be read from that file. --keep-expr will be
applied before --javascript, so the --javascript record
function will not be called for records filtered out by --keep-expr.
See also
For full details of functions available in --keep-expr and --javascript see RTG JavaScript filtering API
Simple filtering by JavaScript expression with --keep-expr¶
The --keep-expr flag aims to provide a convenient way to apply some
simple (typically one line) filtering expressions which are evaluated in
the context of each record. The final expression of the fragment must
evaluate to a boolean value. Records which evaluate to true will be
retained, while false will be removed. The value must be of type
boolean, simply being truthy/falsy (in the JavaScript sense) will raise
an error.
--keep-expr examples:¶
The following expression keeps records where the NA12878 sample has
GQ > 30 and the total depth is > 20. JavaScript will auto convert
numerical strings when comparing a string with a number, so calls to parseInt can be omitted.
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz \
--keep-expr "'NA12878'.GQ > 30 && INFO.DP > 20"
If the field of interest may contain the missing value (‘.’) or may be
entirely missing on a per-record basis, the has() function can be
used to control whether such records are kept vs filtered. For example,
to keep records with depth greater than 20, and remove any without a
DP annotation:
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz \
--keep-expr "has(INFO.DP) && INFO.DP > 20"
Alternatively, to keep records with depth greater than 20, as well as those without a DP annotation:
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz \
--keep-expr "!has(INFO.DP) || INFO.DP > 20"
The next example keeps records where all samples have a depth > 10. The
standard JavaScript array methods every and some can be used to
apply a condition on every sample column.
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz \
--keep-expr "SAMPLES.every(function(s) {return s.DP > 10})"
Similarly, the following example retains records where the FILTER
field is unset, or if set must be either PASS or MED_QUAL:
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz \
--keep-expr "FILTER.every(function(f) {return f == 'PASS' || f == 'MED_QUAL'})"
Note that multi-valued INFO and FORMAT fields are not split into
sub-values, so in some cases correct filtering may require splitting the
values first. For example, to select bi-allelic records with AF
greater than 0.1, the following simple selection will work:
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz \
--keep-expr "INFO.AF>=0.1"
However, in the presence of multi-allelic records, something like the following is required:
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz \
--keep-expr "INFO.AF.split(',').some(function(af) {return af >= 0.1})"
Advanced JavaScript filtering with --javascript¶
The --javascript option aims to support more complicated processing
than --keep-expr, permitting modification of the output VCF, or
supporting use cases where the script is tasked to compute and output
alternative information in addition to (or instead of) the output VCF.
The scripts specified by the user are evaluated once at the start of
processing. Two special functions may be defined in a --javascript
script, which will then be executed in different contexts:
A function with the name
recordwill be executed once for each VCF record. If therecordfunction has a return value it must have type boolean. Records which evaluate totruewill be retained, whilefalsewill be removed. If the record function has no return value then the record will be retained. Therecordfunction is applied after any--keep-exprexpression.A function with the name
endwill be called once at the end of processing. This allows reporting of summary statistics collected during the filter process.
This --javascript flag may be specified multiple times, they will be
evaluated in order, in a shared JavaScript namespace, before VCF
processing commences. This permits a use case where an initial
JavaScript expression supplies parameter values which will be required
by a subsequent JavaScript file.
Example --javascript scripts:¶
To find indels with length greater than 5, save the following to a file
named find-indels.js:
// Finds indels with length > 5
function record() {
var deltas = ALT.map(function (alt) {
return Math.abs(alt.length - REF.length);
});
return deltas.some(function (delta) {return delta > 5});
}
Then perform the filtering via:
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz --javascript find-indels.js
The following example derives a new FORMAT column containing variant
allelic fraction to two decimal places based on the values in the AD
and DP FORMAT annotations, for every sample contained in the
VCF. Save the following to a file named add-vaf.js:
// Derive new VAF FORMAT field for each sample
ensureFormatHeader('##FORMAT=<ID=VAF,Number=1,Type=Float,' +
'Description="Variant Allelic Fraction">');
function record() {
SAMPLES.forEach(function(sample) {
// Take all but the first AD value as numerics
var altDepths = sample.AD.split(",").slice(1);
// Find the max
var maxAltDepth = Math.max.apply(null, altDepths);
if (maxAltDepth > 0) {
sample.VAF = (maxAltDepth / sample.DP).toFixed(2);
}
});
}
Then run the filtering via:
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz --javascript add-vaf.js
The next example produces a table of binned indel lengths, save the
following to a file named indel-lengths.js:
// bin breakpoints can be customised by defining your own bins[] in a
// previous -j flag
if (typeof bins == "undefined") {
var bins = [-10, -5, -3, 0, 4, 6, 11];
}
var counts = [0];
bins.forEach(function () {counts.push(0)});
function record() {
if (ALT.length == 0) {
return false;
}
var deltas = ALT.map(function (alt) { return alt.length - REF.length; });
var maxDel = Math.min.apply(null, deltas);
var maxIns = Math.max.apply(null, deltas);
var delta = Math.abs(maxDel) > maxIns ? maxDel : maxIns;
if (delta == 0) {
return false;
}
for (var i = 0; i < bins.length; i++) {
if (delta < bins[i]) {
counts[i]++;
break;
}
}
if (delta > bins[bins.length - 1]) {
counts[counts.length - 1]++;
}
return false;
}
function end() {
print("Delta\\tCount");
for (var i = 0; i < bins.length; i++) {
print("<" + bins[i] + "\\t" + counts[i]);
}
print(">" + bins[bins.length - 1] + "\\t" + counts[counts.length - 1]);
}
Then run the filtering via:
$ rtg vcffilter -i in.vcf.gz -o out.vcf.gz --javascript indel-lengths.js
We could use this same script with adjusted bins and omitting the output of the VCF via:
$ rtg vcffilter -i in.vcf.gz -j "var bins = [-20, -10, 0, 20, 20];" \
-j indel-lengths.js
See also
For full details of functions available in --keep-expr and --javascript see RTG JavaScript filtering API
See also
snp, family, somatic, population, vcfannotate, vcfmerge, vcfsubset
vcfmerge¶
Synopsis:
Combines the contents of two or more VCF files. The vcfmerge command
can concatenate the outputs of per-chromosome variant detection runs to
create a complete genome VCF file, and also merge VCF outputs from
multiple samples to form a multi-sample VCF file.
Syntax:
$ rtg vcfmerge [OPTION]... -o FILE FILE+
Example:
$ rtg vcfmerge -o merged.vcf.gz snp1.vcf.gz snp2.vcf.gz
Parameters:
File Input/Output |
||
|---|---|---|
|
If set, only read VCF records that overlap the ranges contained in the specified BED file. |
|
|
|
File containing a list of VCF format files (1 per line) to be merged. |
|
|
Output VCF file. Use ‘-’ to write to standard output. |
|
If set, only read VCF records within the specified range. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
|
Input VCF files to merge. May be specified 0 or more times. |
|
Utility |
||
|---|---|---|
|
|
File containing VCF header lines to add, or a literal header line. May be specified 0 or more times. |
|
|
Allow merging of specified header ID even when descriptions do not match. May be specified 0 or more times, or as a comma separated list. |
|
|
Attempt merging of all non-matching header declarations. |
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Prevent VCF header from being written. |
|
|
Do not merge multiple records if the ALTs are different. |
|
|
Do not merge multiple records at the same position into one. |
|
|
Do not merge multiple records containing unmergeable FORMAT fields (Default is to remove those FORMAT fields so the variants can be combined) |
|
|
Output statistics for the merged VCF file. |
|
Usage:
The vcfmerge command takes a list of VCF files and outputs to a
single VCF file. Each VCF file must be block compressed and have a
corresponding tabix index file, which is the default for outputs from
RTG variant detection tools, but may also be created from an existing
VCF file using the RTG bgzip and index commands.
There are two primary usage scenarios for the vcfmerge command. The
first is to combine input VCFs corresponding to different genomic
regions (for example, if variant calling was carried out for each
chromosome independently on different nodes of a compute cluster). The
second scenario is when combining VCFs containing variant calls for
different samples (e.g. combining calls made for separate cohorts into a
single VCF).
The input files must have consistent header lines, although specific
similar header lines can be forced to merge using the --force-merge
parameter, or inconsistent header checking can be entirely disabled with
--force-merge-all.
The --add-header option allows inserting arbitrary VCF header lines
into the output VCF. For more information, see vcfannotate.
Merging records at the same position¶
When multiple records occur with the same position and length on the
reference, vcfmerge will attempt to combine the records into a
single record. Combining multiple fully annotated records is
non-trivial, and can lead to loss of information depending on the
annotations present. The default behavior takes a pragmatic approach to
merging, with options to adjust the merging behavior as
required. Multi-record merging can be disabled entirely by setting
--no-merge-records.
The first point to note is that the QUAL, FILTER, INFO
fields are taken from the first record only (those values from other
records at the position are ignored).
If the combined record would result in a change in the set of ALT
alleles, any VCF INFO or FORMAT fields declared to be of type
A, G, or R cannot meaningfully be retained. By default such
fields will be removed or set to the missing value (.). (Other
annotations may also become semantically meaningless, but it isn’t
possible to tell in general.)
Similarly, if multiple input records with the same position and length on the reference contain information for the same sample, only the information from the first record will be retained.
These behaviors can be altered with additional flags:
--no-merge-alts will simply prevent record merging if the ALTs are
not the same across the records; --preserve-formats will attempt
merging as long as the records do not contain problematic A, G,
or R annotations, or contain the same sample.
See also
snp, family, population, somatic, vcffilter, vcfannotate, vcfsubset, bgzip, index
vcfsplit¶
Synopsis:
Splits samples contained within a VCF into separate files, one per sample.
Syntax:
$ rtg vcfsplit [OPTION]... -i FILE -o DIR
Example:
$ rtg vcfsplit --keep-sample NA12878,NA12891,NA12892 -i population-ceph-calls.vcf.gz -o trio-vcfs
Parameters:
File Input/Output |
||
|---|---|---|
|
If set, only read VCF records that overlap the ranges contained in the specified BED file. |
|
|
|
The input VCF, or ‘-’ to read from standard input. |
|
|
Directory for output. |
|
If set, only read VCF records within the specified range. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
Filtering |
||
|---|---|---|
|
Keep records where the sample is reference. |
|
|
File containing sample IDs to select, or a literal sample name. May be specified 0 or more times, or as a comma separated list. |
|
|
File containing sample IDs to ignore, or a literal sample name. May be specified 0 or more times, or as a comma separated list. |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
Usage:
The vcfsplit command allows producing separate single-sample VCF
files from a single multi-sample VCF, and is more efficient than running
vcfsubset separately for each required sample, particularly when the
input VCF contains many samples, as the input VCF only needs to be read
once.
The output VCFs are all written into the specified output directory, as
sample_name.vcf.gz. For any particular output VCF, only records
where the sample has a non-reference genotype will be output, unless
--keep-ref is used.
See also
vcfstats¶
Synopsis:
Display simple statistics about the contents of a set of VCF files.
Syntax:
$ rtg vcfstats [OPTION]... FILE+
Example:
$ rtg vcfstats /data/human/wgs/NA19240/snp_chr5.vcf.gz
Location : /data/human/wgs/NA19240/snp_chr5.vcf.gz
Passed Filters : 283144
Failed Filters : 83568
SNPs : 241595
MNPs : 5654
Insertions : 15424
Deletions : 14667
Indels : 1477
Unchanged : 4327
SNP Transitions/Transversions : 1.93 (210572/108835)
Total Het/Hom ratio : 2.13 (189645/89172)
SNP Het/Hom ratio : 2.12 (164111/77484)
MNP Het/Hom ratio : 3.72 (4457/1197)
Insertion Het/Hom ratio : 1.69 (9695/5729)
Deletion Het/Hom ratio : 2.33 (10263/4404)
Indel Het/Hom ratio : 3.13 (1119/358)
Insertion/Deletion ratio : 1.05 (15424/14667)
Indel/SNP+MNP ratio : 0.13 (31568/247249)
Parameters:
File Input/Output |
||
|---|---|---|
|
Set to only calculate statistics for known variants. |
|
|
Set to only calculate statistics for novel variants. |
|
|
Set to only calculate statistics for the specified sample. (Default is to include all samples). May be specified 0 or more times. |
|
FILE+ |
VCF file from which to derive statistics. Use ‘-’ to read from standard input. Must be specified 1 or more times. |
|
Reporting |
||
|---|---|---|
|
Set to output variant length histogram. |
|
Utility |
||
|---|---|---|
|
|
Prints help on command-line flag usage. |
Usage:
Use the vcfstats command to display summary statistics for a set of
VCF files. If a VCF file contains multiple sample columns, the
statistics for each sample are shown individually.
When determining the categorization of a REF to ALT transformation, some
normalization is carried out to ignore same as reference bases at the
start and end of the alleles. Thus the following REF to ALT
transformations are categorized as SNPs:
A -> G (simple case)
ATGC -> ATGG (leading bases match)
ATGC -> ACGC (leading and trailing bases match)
Cases where multiple bases change, but the lengths of the two alleles do
not are considered to be MNPs:
ATGC -> TTGG (two bases change)
ATGC -> GTCT (three bases change)
Cases where there is pure addition or removal of bases are classified as
Insertions or Deletions respectively:
A -> AT (one base insertion)
ATT -> ATTTT (two base insertion)
AT -> A (one base deletion)
ATTTT -> ATT (two base deletion)
The remaining case is there there is a length change between the REF and
ALT, but it is not pure. These are called Indels:
ATT -> CTTT (one base changed, one base inserted)
CTTT -> ATT (one base changed, one base deleted)
In the per-sample summary output of vcfstats, each genotype is
classified as a whole into one of the above categories, preferring the
more complex of the transformations when ploidy is greater than one.
When computing the per-sample variant length histograms, note that the histograms are incremented for each called allele (thus a diploid homozygous call will increment the appropriate cell by two), and the length of an indel is taken as the change in length rather than the overall length.
vcfsubset¶
Synopsis:
Create a VCF file containing a subset of the original columns.
Syntax:
$ rtg vcfsubset [OPTION]... -i FILE -o FILE
Example:
$ rtg vcfsubset -i snps.vcf.gz -o frequency.vcf.gz --keep-info AF --remove-samples
Parameters:
File Input/Output |
||
|---|---|---|
|
If set, only read VCF records that overlap the ranges contained in the specified BED file. |
|
|
|
VCF file containing variants to manipulate. Use ‘-’ to read from standard input. |
|
|
Output VCF file. Use ‘-’ to write to standard output. |
|
If set, only read VCF records within the specified range. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
Filtering |
||
|---|---|---|
|
Keep the specified FILTER tag. May be specified 0 or more times, or as a comma separated list. |
|
|
Keep the specified FORMAT field. May be specified 0 or more times, or as a comma separated list. |
|
|
Keep the specified INFO tag. May be specified 0 or more times, or as a comma separated list. |
|
|
File containing sample IDs to keep, or a literal sample name. May be specified 0 or more times, or as a comma separated list. |
|
|
Remove the specified FILTER tag. May be specified 0 or more times, or as a comma separated list. |
|
|
Remove all FILTER tags. |
|
|
Remove the specified FORMAT field. May be specified 0 or more times, or as a comma separated list. |
|
|
Remove the contents of the ID field. |
|
|
Remove the specified INFO tag. May be specified 0 or more times, or as a comma separated list. |
|
|
Remove all INFO tags. |
|
|
Remove the QUAL field. |
|
|
File containing sample IDs to remove, or a literal sample name. May be specified 0 or more times, or as a comma separated list. |
|
|
Remove all samples. |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Prevent VCF header from being written. |
|
Usage:
Use the vcfsubset command to produce a smaller copy of an original VCF
file containing only the columns and information desired. For example,
to produce a VCF containing only the information for one sample from a
multiple sample VCF file use the --keep-sample flag to specify the
sample to keep. The various --keep and --remove options can either be
specified multiple times or with comma separated lists, for example,
--keep-format GT --keep-format DP is equivalent to –keep-format GT,DP.
See also
vcf2rocplot¶
Synopsis:
Produce rocplot compatible ROC data files from vcfeval annotated
VCFs. The primary use cases for this command are to produce aggregate
ROCs from several independent vcfeval evaluation runs, and to
generate ROCs with respect to different criteria than were used during
the initial vcfeval evaluation.
Syntax:
$ rtg vcf2rocplot [OPTION]... -o DIR FILE+
Create an aggregate ROC from evaluations of several independent samples:
$ rtg vcfeval -b goldstandard.vcf.gz -c snps.vcf.gz -t HUMAN_reference \
--sample daughter1 -f AVR -o eval -m annotate
$ rtg vcfeval -b goldstandard.vcf.gz -c snps.vcf.gz -t HUMAN_reference \
--sample daughter2 -f AVR -o eval -m annotate
$ rtg vcfeval -b goldstandard.vcf.gz -c snps.vcf.gz -t HUMAN_reference \
--sample son2 -f AVR -o eval -m annotate
$ rtg vcf2rocplot -o eval_family -f AVR \
eval_{daughter1,daughter2,son1}/{baseline,calls}.vcf.gz
$ rtg rocplot eval_family/weighted_roc.tsv.gz \
--png eval_family_roc.png
Parameters:
File Input/Output |
||
|---|---|---|
|
If set, only read VCF records that overlap the ranges contained in the specified BED file. |
|
|
|
Directory for output. |
|
If set, only read VCF records within the specified range. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
|
Input VCF files containing vcfeval annotations. Must be specified 1 or more times. |
|
Reporting |
||
|---|---|---|
|
Output summary statistics where precision >= supplied value (Default is to summarize at maximum F-measure) |
|
|
Output summary statistics where score meets supplied value (Default is to summarize at maximum F-measure) |
|
|
Output summary statistics where sensitivity >= supplied value (Default is to summarize at maximum F-measure) |
|
|
Output ROC files for each intersection of –roc-subset, –roc-expr, and –roc-regions values provided. |
|
|
Output ROC file for variants matching custom JavaScript expression. Use the form <LABEL>=<EXPRESSION>. May be specified 0 or more times. |
|
|
Output ROC file for variants overlapping custom regions supplied in BED file. Use the form <LABEL>=<FILENAME>. May be specified 0 or more times. |
|
|
Output ROC file for preset variant subset. Allowed values are [hom, het, snp, non-snp, mnp, indel, dup, del]. May be specified 0 or more times, or as a comma separated list. |
|
|
|
The order in which to sort the ROC scores so that |
|
|
The name of the VCF FORMAT field to use as the ROC score. Also valid are |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
|
Number of threads (Default is the number of available cores) |
Usage:
While the ROC outputs generated by vcfeval/cnveval/bndeval are
sufficient for many scenarios, there are some situations where it is useful
to regenerate ROC files from one or more existing vcfeval outputs.
One such situation is when evaluating caller accuracy across a cohort of samples where the number of variants per individual sample is low. This is a particular problem with CNV calling and restricted gene panels. Individual sample ROC curves are fairly uninformative with regard to overall accuracy or where to place suitable filter thresholds. A ROC curve from the combined evaluations provides a much better indication of overall caller accuracy.
Another use case for generating ROC files from an existing evaluation
run is to look at alternative scoring methods or stratifications without
having to execute the time-consuming variant matching stage implied by a
new vcfeval run.
The prerequisite for using vcf2rocplot with vcfeval outputs is that
vcfeval must have been run in “annotation” mode (i.e. via
--output-mode=annotate) so that the output VCF files contain sufficient
annotations regarding variant classification status. It is important that
vcf2rocplot be provided with both annotated baseline and call VCFs in
order to produce correct outputs.
The operation of the various score field selection and ROC
stratification flags works the same as when running vcfeval.
vcf2rocplot is compatible with the outputs of vcfeval, cnveval
and bndeval, although these should not be mixed in a single run.
svdecompose¶
Synopsis:
Split composite structural variants into a breakend representation.
Syntax:
$ rtg svdecompose [OPTION]... -i FILE -o FILE
Parameters:
File Input/Output |
||
|---|---|---|
|
|
VCF file containing variants to filter. Use ‘-’ to read from standard input. |
|
|
Output VCF file name. Use ‘-’ to write to standard output. |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
Prevent VCF header from being written. |
|
Usage:
The svdecompose command is applied to a VCF containing structural
variants and converts deletion, insertion, inversion, and tandem
duplications with SVTYPE of DEL, INS, INV, and
DUP, respectively, into corresponding breakend events with
SVTYPE=BND. svdecompose will also decompose sequence-resolved
insertions and deletions greater than --min-indel-length into
breakend representation. Records of others types are passed through
without modification.
This operation can be useful for the purposes of reducing output from
various structural variant callers to a common representation to
better facilitate comparison with the bndeval command.
For insertions, svdecompose will represent the insertion as
breakends between the reference and a “virtual haplotype”, where for
example, contig “<INS_A>” represents the destination of all insertions
made on chromosome A. So if another caller produced a similar insertion
event (in position and/or length), the break end versions will also be
nearby on the virtual contig. For the following insertions:
1 54712 . T TTTTTTTTTCTTTCTTTCTTTCTTTCTTTCTTTCTTTCTTTCTTTCTTTCTTTC . . .
1 934144 I_7 C CGGAGGGGAGGGCGCGGAGCGGAGG . . .
1 934144 I_22 C CGGAGGGGAGGGCGCGGAGCGGAGGGGAGGGCGCGGAGCGGAGG . . .
each insertion gets two breakends like this:
1 54712 . T T[<INS_1>:54712[ . . SVTYPE=BND;CIPOS=0,0
1 54712 . T ]<INS_1>:54765]C . . SVTYPE=BND;CIPOS=0,0
1 934144 I_7 C C[<INS_1>:934144[ . . SVTYPE=BND;CIPOS=0,0
1 934144 I_22 C C[<INS_1>:934144[ . . SVTYPE=BND;CIPOS=0,0
1 934144 I_7 C ]<INS_1>:934168]G . . SVTYPE=BND;CIPOS=0,0
1 934144 I_22 C ]<INS_1>:934187]G . . SVTYPE=BND;CIPOS=0,0
See also
bndeval¶
Synopsis:
Evaluate called breakends for agreement with a baseline breakend set.
Outputs a weighted ROC file which can be viewed with rtg rocplot
and VCF files containing false positives (called breakends not matched
in the baseline), false negatives (baseline breakends not matched in
the call set), and true positives (breakends that match between the
baseline and calls).
Syntax:
$ rtg bndeval [OPTION]... -b FILE -c FILE -o DIR
Parameters:
File Input/Output |
||
|---|---|---|
|
|
VCF file containing baseline variants. |
|
If set, only read VCF records that overlap the ranges contained in the specified BED file. |
|
|
|
VCF file containing called variants. |
|
|
Directory for output. |
|
If set, only read VCF records within the specified range. The format is one of <sequence_name>, <sequence_name>:<start>-<end>, <sequence_name>:<pos>+<length> or <sequence_name>:<pos>~<padding> |
|
Filtering |
||
|---|---|---|
|
Use all records regardless of FILTER status (Default is to only process records where FILTER is “.” or “PASS”) |
|
|
If set, allow matches between flipped breakends. |
|
|
Positional tolerance for breakend matching (Default is 100) |
|
Reporting |
||
|---|---|---|
|
Do not produce ROCs. |
|
|
|
Output reporting mode. Allowed values are [split, annotate] (Default is split) |
|
|
The order in which to sort the ROC scores so that |
|
|
The name of the VCF FORMAT field to use as the ROC score. Also valid are |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
Usage:
The bndeval command operates on VCF files containing breakends such
as those produced by the discord command. In particular, it
considers records having the breakend structural variant type
(SVTYPE=BND) as defined in the VCF specification. Other types of
record are ignored, but the svdecompose command can be applied
beforehand to split certain other structural variants (e.g., INV and
DEL) or sequence-resolved insertions and deletions into constituent
breakend events.
The input and output requirements of bndeval are broadly similar
to the vcfeval command. The primary inputs to bndeval are a
truth/baseline VCF containing expected breakends, and a query/call VCF
containing the called breakends. Evaluation can be restricted to
particular regions by specifying a BED file.
The regions contained in the evaluation regions BED file are intersected
with the breakend records contained in the truth VCF in order to obtain a
list of truth breakend regions. An evaluation region is included if there
is any overlapping truth VCF record (no attempt is made to look at the
degree of overlap). Thus by supplying either evaluation regions
corresponding to targeted regions or larger gene-level regions bndeval
can be used to evaluate at different levels of granularity.
Similarly, the evaluation regions are intersected with the breakend records records contained in the calls VCF to obtain called breakend regions.
The truth breakend regions are then intersected with the called breakend
regions to obtain TP/FP/FN metrics. The intersection supports a
user-selectable tolerance in position. Further, by default, a breakend
must occur in the same orientation to be considered a match, but this
constraint can be relaxed by supplying the --bidirectional command
line option.
As with vcfeval, a selection of ROC files can be generated according to various score field selection and ROC stratification flags. See vcfeval for more information.
bndeval outputs¶
Once complete, bndeval command produces summary statistics and the
following primary result files in the output directory:
weighted_roc.tsv.gz- contains ROC data that can be plotted withrocplotbaseline.bed.gzcontains the truth breakend regions, where each BED record contains the region status asTPorFN, theSVTYPE, and the span of the original truth VCF record.calls.bed.gzcontains the called breakend regions, where each BED record contains the region status asTPorFP, theSVTYPE, the span of the original calls VCF record, and the score value used for ranking in the ROC plot.summary.txtcontains the same summary statistics printed to standard output.
See also
discord, svdecompose, vcfeval, cnveval, vcf2rocplot, rocplot
cnveval¶
Synopsis:
Evaluate called CNV regions for agreement with a baseline CNV set and
output ROC information in a form compatible with rtg rocplot.
Syntax:
$ rtg cnveval [OPTION]... -b FILE -c FILE -e FILE -o DIR
Parameters:
File Input/Output |
||
|---|---|---|
|
|
VCF file containing baseline variants. |
|
|
VCF file containing called variants. |
|
|
Evaluate with respect to the supplied regions of interest BED file. |
|
|
Directory for output. |
Filtering |
||
|---|---|---|
|
Use all records regardless of FILTER status (Default is to only process records where FILTER is “.” or “PASS”) |
|
Reporting |
||
|---|---|---|
|
Output summary statistics where precision >= supplied value (Default is to summarize at maximum F-measure) |
|
|
Output summary statistics where score meets supplied value (Default is to summarize at maximum F-measure) |
|
|
Output summary statistics where sensitivity >= supplied value (Default is to summarize at maximum F-measure) |
|
|
Include evaluation region names in annotated outputs. |
|
|
Do not produce ROCs. |
|
|
Output ROC files for each intersection of –roc-subset, –roc-expr, and –roc-regions values provided. |
|
|
Output ROC file for variants matching custom JavaScript expression. Use the form <LABEL>=<EXPRESSION>. May be specified 0 or more times. |
|
|
Output ROC file for variants overlapping custom regions supplied in BED file. Use the form <LABEL>=<FILENAME>. May be specified 0 or more times. |
|
|
Output ROC file for preset variant subset. Allowed values are [dup, del]. May be specified 0 or more times, or as a comma separated list. |
|
|
|
The order in which to sort the ROC scores so that |
|
|
The name of the VCF FORMAT field to use as the ROC score. Also valid are |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
|
|
Do not gzip the output. |
|
|
Number of threads (Default is the number of available cores) |
Usage:
The cnveval command operates on the CNV VCF files that are produced
by segment, but in principal could be used with any VCF file that
follows the VCF specification for structural variants. The VCF records
must contain an INFO END annotation, and INFO SVTYPE
annotation containing a value of either DUP or DEL.
The primary inputs to cnveval are an evaluation regions BED file, a
truth/baseline VCF containing expected CNV calls, and a query/calls VCF
containing the called CNV regions.
The regions contained in the evaluation regions BED file are intersected
with the CNV records contained in the truth VCF in order to obtain a list
of truth CNV regions, which each have a status of DUP or DEL. An
evaluation region is included if there is any overlapping truth VCF record
(no attempt is made to look at the degree of overlap). Thus by supplying
either evaluation regions corresponding to targeted regions or larger
gene-level regions cnveval can be used to evaluate at different levels
of granularity.
Similarly, the evaluation regions are intersected with the CNV records records contained in the calls VCF to obtain called CNV regions.
The truth CNV regions are then intersected with the called CNV regions to obtain TP/FP/FN metrics.
As with vcfeval, a selection of ROC files can be generated according to various score field selection and ROC stratification flags. See vcfeval for more information.
cnveval outputs¶
Once complete, cnveval command produces summary statistics and the
following primary result files in the output directory:
weighted_roc.tsv.gz- contains ROC data that can be plotted withrocplotbaseline.bed.gzcontains the truth CNV regions, where each BED record contains the region status asTPorFN, theSVTYPE, and the span of the original truth VCF record.calls.bed.gzcontains the called CNV regions, where each BED record contains the region status asTPorFP, theSVTYPE, the span of the original calls VCF record, and the score value used for ranking in the ROC plot.summary.txtcontains the same summary statistics printed to standard output.
Evaluation per target region¶
The default method of evaluation is with respect to target regions, where each individual region of interest will be evaluated as correct or incorrect. For this mode, supply the standard target regions BED file, and both VCF files.
For RTG calls, directly supply either the raw VCF produced by
segment or after filtering with vcffilter
Evaluation per gene region¶
A second form of evaluation is when the desired level of granularity is
at the gene level. For this mode, the evaluation regions should be one
region per gene (this is the same BED file that is used with
--summary-regions when running segment or cnvsummary), and
the input VCFs should be pre-processed to contain at most one record per
gene.
See also
segment, cnvsummary, vcffilter, vcfeval, bndeval, vcf2rocplot, rocplot
pedfilter¶
Synopsis:
Filter and convert a pedigree file.
Syntax:
$ rtg pedfilter [OPTION]... FILE
Example:
$ rtg pedfilter --remove-parentage mypedigree.ped
Parameters:
File Input/Output |
||
|---|---|---|
|
The pedigree file to process, may be PED or VCF, use ‘-’ to read from stdin. |
|
Filtering |
||
|---|---|---|
|
Keep only individuals with the specified family ID. May be specified 0 or more times, or as a comma separated list. |
|
|
Keep only individuals with the specified ID. May be specified 0 or more times, or as a comma separated list. |
|
|
Keep only primary individuals (those with a PED individual line / VCF sample column) |
|
|
Remove all parent-child relationship information. |
|
Reporting |
||
|---|---|---|
|
Output pedigree in in the form of a VCF header rather than PED. |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
Usage:
The pedfilter command can be used to perform manipulations on pedigree
information and convert pedigree information between PED and VCF header
format. For more information about the PED file format see Pedigree PED input file format.
The VCF files output by the family and population commands contain
full pedigree information represented as VCF header lines, and the
pedfilter command allows this information to be extracted in PED
format.
This command produces the pedigree output on standard output, which can be redirected to a file or another pipeline command as required.
See also
pedstats¶
Synopsis:
Output information from pedigree files of various formats.
Syntax:
$ rtg pedstats [OPTION]... FILE
Example:
For a summary of pedigree information:
$ rtg pedstats ceph_pedigree.ped
Pedigree file: /data/ceph/ceph_pedigree.ped
Total samples: 17
Primary samples: 17
Male samples: 9
Female samples: 8
Afflicted samples: 0
Founder samples: 4
Parent-child relationships: 26
Other relationships: 0
Families: 3
To output a list of all founders:
$ rtg pedstats --founder-ids ceph_pedigree.ped
NA12889
NA12890
NA12891
NA12892
For quick pedigree visualization using Graphviz and ImageMagick, use a command-line such as:
$ dot -Tpng <(rtg pedstats --dot "A Title" mypedigree.ped) | display -
Parameters:
File Input/Output |
||
|---|---|---|
|
The pedigree file to process, may be PED or VCF, use ‘-’ to read from stdin. |
|
Reporting |
||
|---|---|---|
|
|
Output id lists using this separator (Default is \n) |
|
Output pedigree in Graphviz format, using the supplied text as a title. |
|
|
Output information about family structures. |
|
|
Output ids of all females. |
|
|
Output ids of all founders. |
|
|
Output ids of all males. |
|
|
Output ids of maternal individuals. |
|
|
Output ids of paternal individuals. |
|
|
Output ids of all primary individuals. |
|
|
When outputting Graphviz format, use a layout that looks less like a traditional pedigree diagram but works better with large complex pedigrees. |
|
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
Usage:
This command is used to show pedigree summary statistics or select groups of individual IDs.
When using pedstats to output a list of sample IDs, the default is
to print one ID per line. Depending on subsequent use, it may be
convenient to use a different separator between output IDs. For example,
with comma separated output it is possible to directly use the results
as an argument to vcfsubset:
$ rtg vcfsubset -i pedigree-calls.vcf.gz -o family1.vcf.gz \
--keep-samples <(rtg pedstats -d , --founder-ids ceph_pedigree.ped)
In addition, pedstats can be used to generate a simple pedigree
visualization, using the well-known Graphviz graphics drawing package,
which can be saved to PNG or PDF. For example, with the following
chinese-trio.ped:
#PED format pedigree
#
#fam-id/ind-id/pat-id/mat-id: 0=unknown
#sex: 1=male; 2=female; 0=unknown
#phenotype: -9=missing, 0=missing; 1=unaffected; 2=affected
#
#fam-id ind-id pat-id mat-id sex phen
0 NA24631 NA24694 NA24695 1 0
0 NA24694 0 0 1 0
0 NA24695 0 0 2 0
We can visualize the pedigree with:
$ dot -Tpng <(rtg pedstats --dot "Chinese Trio" chinese-trio.ped) -o chinese-trio.png
This will create a PNG image that can be displayed in any image viewing tool and contains the pedigree structure as shown below.
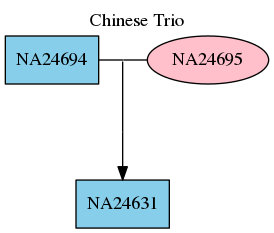
For more information about the PED file format see Pedigree PED input file format.
The VCF files output by the RTG pedigree-aware variant calling commands
contain full pedigree information represented as VCF header lines, and
the pedstats command can also take these VCFs as input. For example,
given a VCF produced by the population command after calling the
CEPH-1463 pedigree:
$ dot -Tpng <(rtg pedstats --dot "CEPH 1463" population-ceph-calls.vcf.gz) -o ceph-1463.png
Would produce the following pedigree directly from the VCF:
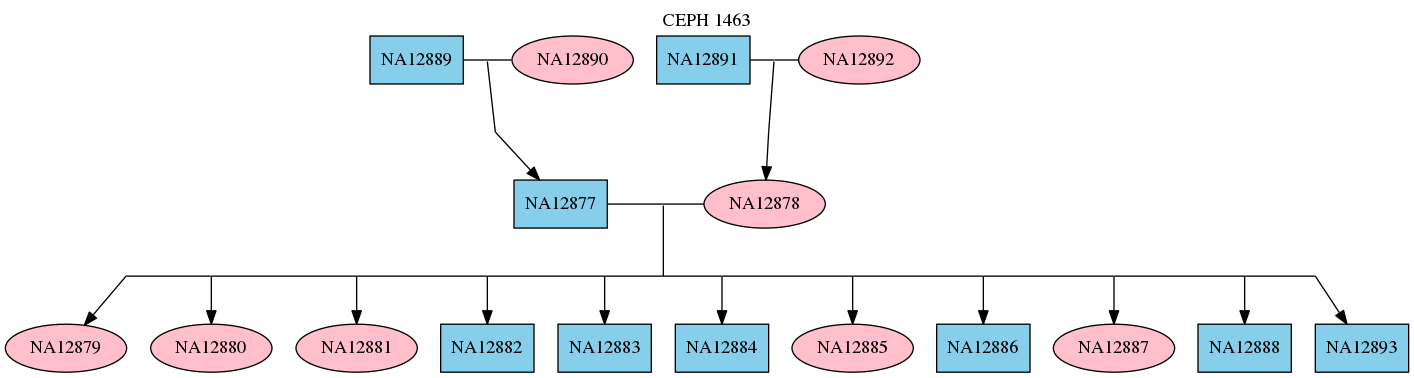
Note
Graphviz is provided directly via many operating system package managers, and can also be downloaded from their web site: https://www.graphviz.org/
See also
rocplot¶
Synopsis:
Plot ROC curves from readsimeval and vcfeval ROC data files, either
to an image, or using an interactive GUI.
Syntax:
$ rtg rocplot [OPTION]... FILE+
$ rtg rocplot [OPTION]... --curve STRING
Example:
$ rtg rocplot eval/weighted_roc.tsv.gz
Parameters:
File Input/Output |
||
|---|---|---|
|
ROC data file with title optionally specified (path[=title]). May be specified 0 or more times. |
|
|
If set, output a PNG image to the given file. |
|
|
If set, output a SVG image to the given file. |
|
|
Show a zoomed view with the given coordinates, supplied in the form <xmax>,<ymax> or <xmin>,<ymin>,<xmax>,<ymax> |
|
|
ROC data file. May be specified 0 or more times. |
|
Reporting |
||
|---|---|---|
|
If set, print rocplot command used in previously saved image. |
|
|
If set, hide the side pane from the GUI on startup. |
|
|
If set, interpolate curves at regular intervals. |
|
|
Sets the plot line width (Default is 2) |
|
|
Name of color palette to use. Allowed values are [blind_13, blind_15, blind_8, brewer_accent, brewer_dark2, brewer_paired, brewer_pastel1, brewer_pastel2, brewer_set1, brewer_set2, brewer_set3, classic] (Default is classic) |
|
|
If set, use a plain plot style. |
|
|
If set, show points on each curve. |
|
|
|
If set, plot precision vs sensitivity rather than ROC. |
|
If set, show scores on the plot. |
|
|
|
Title for the plot. |
Utility |
||
|---|---|---|
|
|
Print help on command-line flag usage. |
Usage:
Used to produce ROC plots from the ROC files produced by
readsimeval, bndeval and vcfeval. By default this opens the
ROC plots in an interactive viewer. On a system with only console access
the plot can be saved directly to an image file using the either the
--png or --svg parameter.
ROC data files may be specified either as direct file arguments to the
command, or via the --curve flag. The former method is useful when
selecting files using shell wild card globbing, and the latter method
allows specifying a custom title for each curve, so use whichever method
is most convenient.
Strictly speaking, a true ROC curve should use rates rather than absolute numbers on the X and Y axes (e.g. True Positive / Total Positives rather than True Positives on the Y, and False Positive / Total Negatives on the X axis). However, there are a couple of difficulties involved with computing these rates with variant calling datasets. Firstly, the truth sets do not include any indication of the set of negatives (the closest we may get is in the cases of truth sets which contain a set of confidence regions, where it can be assumed that no other variants may be present inside the specified regions); secondly even with knowledge of negative regions, how do you count the set of possible negative calls, when a call could occupy multiple reference bases, or even (in the case of insertions) zero reference bases. It is conceptually even possible to have a call-set contain more false positives than there are reference bases. For this reason the ROC curves are plotted using the absolute counts.
Precision/sensitivity (also known as precision/recall) curves are
another popular means of visualizing call-set accuracy, and these
metrics also do not require a count of Total Negatives and so cause no
particular difficulty to plot. Precision/sensitivity graphs can be
selected from the command line via the --precision-sensitivity flag,
or may be interactively selected in the GUI.
An interesting result of ROC analysis is that although there may be few
data points on an ROC graph, it is possible to construct a filtered
dataset corresponding to any point that lies on a straight line between
two points on the graph. (For example, using threshold A for 25% of the
variants and threshold B for 75% of the variants would result in
accuracy that is 75% of the way between the points corresponding to
thresholds A and B on the ROC plot). So in a sense it is meaningful to
connect points on an ROC graph with straight lines. However, for
precision/sensitivity graphs, it’s incorrect to connect adjacent points
with a straight line, as this does not correspond to achievable accuracy
on the ROC convex hull and can over-estimate the accuracy. Instead, we
can plot appropriately interpolated values with the --interpolate
option.
The default ROC graphs include some flourishes such as background
gradients and axes drop shadows, these can be disabled via the
--plain parameter. Alternative preset color palettes for the curve
colors may be selected with the --palette parameter, and in
particular some palettes are more color-blind friendly than the default
palette. In addition, PNG images saved by rocplot include metadata
indicating the graph configuration that may be useful when recreating
graphs. This metadata can be displayed (when present) via the --cmd
parameter.
Interactive GUI¶
The following image shows the rocplot GUI with an example ROC plot :
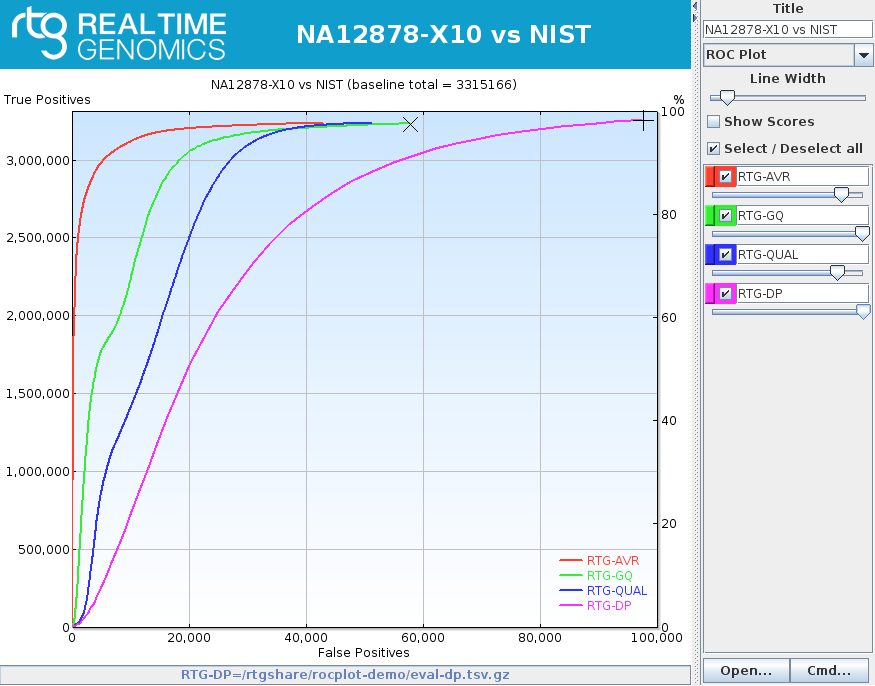
Similarly, here is an example precision/sensitivity plot:
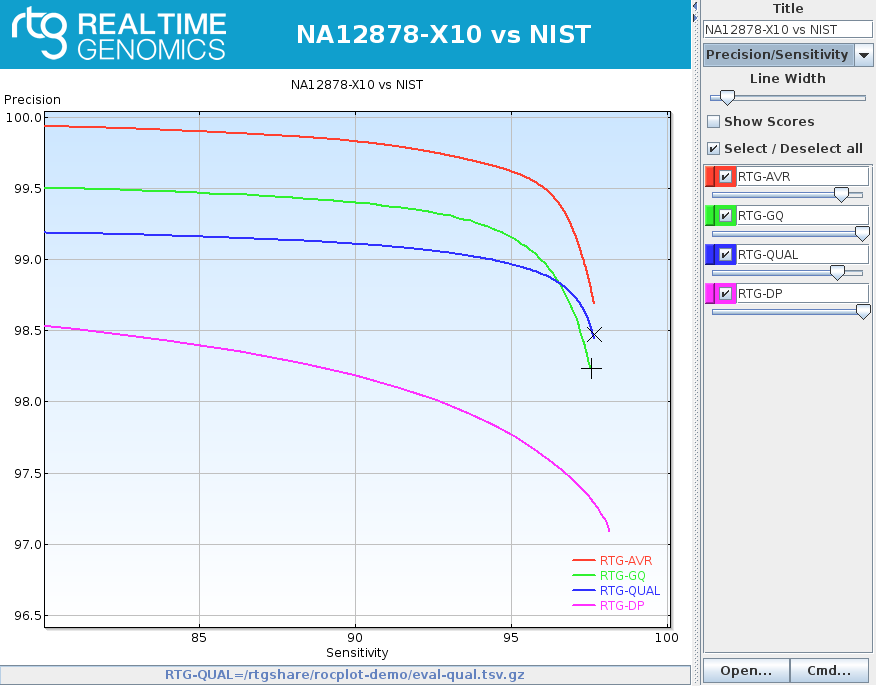
Some quick tips for the interactive GUI:
Select regions within the graph to zoom in. Right click within the graph area to bring up a context menu that allows undoing the zoom one level at a time, or resetting the zoom to the default.
The graph right click menu also allows exporting the image as PNG or SVG. (The saved image does not include the RTG banner).
Click on a spot in the graph to show the equivalent accuracy metrics for that location in the status bar. Clicking to the left or below the axes will remove the cross-hair. Note that sensitivity depends on the baseline total number of variants being correct. If for example the ROC curve corresponds to evaluating an exome call-set against a whole-genome baseline, this number will be inaccurate.
A secondary cross-hair is also available by holding down shift when placing (or removing) the cross-hair. When two cross-hairs are placed or moved, metrics in the status bar indicate the difference between the two positions.
Additional ROC data files can be loaded by clicking on the “Open…” button, and multiple ROC data files within a directory can be loaded at once using multi-select. Alternatively, you may use Drag and Drop from your file browser to drop ROC data files into either the graph area or the right hand ROC curve widget area.
The “Cmd” button will open a message window that contains a command-line equivalent to the currently displayed set of curves. This command-line may be copy-pasted, providing an easy way to replicate the current set of curves in another session, generate a curve in a script, or share with a colleague.
There is a drop down that allows for switching between ROC and precision/sensitivity graph types.
Each curve in the GUI has a customization widget on the right hand side of the window that allows several operations:
Rename the title used for the curve via the editable text.
Temporarily hide/show the curve via selection checkbox.
Reorder curves via drag and drop using the colored handle on the left.
Right click within the ROC widget area to bring up a context menu that allows permanently removing that curve, or customizing the color used for the curve
Each curve has a slider to simulate the effect of applying a threshold on the scoring attribute. If the “show scores” option is set, this provides an easy way to select appropriate filter threshold values, which you might apply to variant sets using
rtg vcffilteror similar VCF filtering tools.
Note
For definitions of the terminology used when evaluating caller accuracy, see: https://en.wikipedia.org/wiki/Receiver_operating_characteristic and https://en.wikipedia.org/wiki/Sensitivity_and_specificity
Note
For a description of the precision/sensitivity interpolation, see: “The relationship between Precision-Recall and ROC curves”, Davis, J., (2006), https://dx.doi.org/10.1145/1143844.1143874
See also
version¶
Synopsis:
The RTG version display utility.
Syntax:
$ rtg version
Example:
$ rtg version
Product: RTG Core 3.9
Core Version: 718f8317b7 (2018-05-29)
RAM: 25.0GB of 31.3GB RAM can be used by rtg (79%)
CPU: Defaulting to 4 of 4 available processors (100%)
JVM: Java HotSpot(TM) 64-Bit Server VM 1.8.0_161
License: Expires on 2019-05-20
Contact: support@realtimegenomics.com
Patents / Patents pending:
US: 7,640,256, 9,165,253, 13/129,329, 13/681,046, 13/681,215, 13/848,653, 13/925,704, 14/015,295, 13/971,654, 13/971,630, 14/564,810
UK: 1222923.3, 1222921.7, 1304502.6, 1311209.9, 1314888.7, 1314908.3
New Zealand: 626777, 626783, 615491, 614897, 614560
Australia: 2005255348, Singapore: 128254
Citation (variant calling):
John G. Cleary, Ross Braithwaite, Kurt Gaastra, Brian S. Hilbush, Stuart Inglis, Sean A. Irvine, Alan Jackson, Richard Littin, Sahar Nohzadeh-Malakshah, Mehul Rathod, David Ware, Len Trigg, and Francisco M. De La Vega. "Joint Variant and De Novo Mutation Identification on Pedigrees from High-Throughput Sequencing Data." Journal of Computational Biology. June 2014, 21(6): 405-419. doi:10.1089/cmb.2014.0029.
Citation (vcfeval):
John G. Cleary, Ross Braithwaite, Kurt Gaastra, Brian S. Hilbush, Stuart Inglis, Sean A. Irvine, Alan Jackson, Richard Littin, Mehul Rathod, David Ware, Justin M. Zook, Len Trigg, and Francisco M. De La Vega. "Comparing Variant Call Files for Performance Benchmarking of Next-Generation Sequencing Variant Calling Pipelines." bioRxiv, 2015. doi:10.1101/023754.
(c) Real Time Genomics, 2025
Parameters:
There are no options associated with the version command.
Usage:
Use the version command to display release and version information.
license¶
Synopsis:
The RTG licensed commands display utility.
Syntax:
$ rtg license
Example:
$ rtg license
Parameters:
There are no options associated with the license command.
Usage:
Use the license command to display the release status of each licensed
command. Output at the command line (standard output) shows command name,
licensed status, and command release level.
help¶
Synopsis:
The RTG help command provides online help for all RTG commands.
Syntax:
List all commands:
$ rtg help
Show usage syntax and flags for one command:
$ rtg help COMMAND
Example:
$ rtg help format
Parameters:
There are no options associated with the help command.
Usage:
Use the help command to view syntax and usage information for the main
rtg command as well as individual RTG commands.
